N. Shiraishi1, Y. Suzuki2, T. Hirose2, S. Jeong3, T. Shimada4, K. Okada5, M. Kuzuya6
1. Department of Rehabilitation, Faculty of Health Science, Nihon Fukushi University; 2. Department of Comprehensive Community Care Systems, Nagoya University Graduate School of Medicine; 3. Department of Social Science, National Center for Geriatrics and Gerontology; 4. Department of rehabilitation, Mie university hospital; 5. Department of Nutritional Sciences, Nagoya University of Arts and Sciences; 6. Department of Community Healthcare & Geriatrics, Nagoya University Graduate School of Medicine
Corresponding Author: Nariaki Shiraishi, Department of Rehabilitation, Faculty of Health Science, Nihon Fukushi University, Japan, n-shira@n-fukushi.ac.jp, Tel +81 569-20-0118 (2326), Fax +81 569-20-0127
Abstract
Objective: To date, the actual prevalence of Skeletal muscle mass (SMM) loss by rigorous definition and its related factors have not been sufficiently surveyed in the community. We therefore examined the factors related to the reductions of skeletal muscle mass (SMM) in older adults. Design: Case-control study. Subjects: One hundred twenty four community-dwelling older adults aged ≥65 years participated. Measurements: Reductions of SMM were assessed by measuring difference between SMM at baseline and SMM 1 year later, by which participants were divided into three groups. Variables of the first tertile group, who had the greatest decrease in SMM, were compared with those of the second/third tertile groups. Variables included hight, weight, body mass index (BMI), maximal knee extension strength, grip strength, lower and upper muscle quality (UMQ), 5-m walking time (WT), timed up and go (TUG), food frequency questionnaire, mini nutritional assessment short form (MNA-SF), basic health checklist. A logistic regression analysis and classification and regression trees (CART) were used for multivariate analysis in order to extract variables that predicted reductions of SMM. Results: Significant differences were observed for age, SMM, UMQ, TUG, and WT between the first tertile and the second/third tertile groups, The CART analysis indicated that vitamin D intake UMQ and 5-m WT predicted significant decrease in SMM. Conclusion: The present study suggested a possibility that future reductions of SMM could be predicted by simple indices that may contribute to early detection of individuals at risk of developing sarcopenia in old age.
Key words: Skeletal muscle mass, older adults, classification and regression tree.
Introduction
With unprecedentedly rapid increase of aged population (1), how older people maintain both physical and mental capacities had become a matter of global concern in aged societies worldwide. It has been believed that activities of daily living (ADL) capabilities decrease with age especially among those aged 75 years or older, approximately 20%–30% of which are frail individuals requiring some kind of support in their daily lives (2). The number of older individuals aged 65 years or older who require care or support provided within the long-term nursing care insurance system was 4.378 million in the fiscal year 2007, which accounted for 15.9% of entire aged populationand was an increase of 1.501 million compared with the figure reported in the fiscal year 2001 (3). In view of prophylaxis, early detection of individuals at risk of developing geriatric syndromes, such as falls, incontinence, malnutrition, degraded lifestyles, depression, and dementia, are considered important for maintaining quality of life and reducing social security costs for older population. According to the comprehensive survey of living conditions implemented nationwide, a decline in muscle strength and muscle mass leading to muscular weakening accounted for the cause of requiring support and care in approximately 30% and 20% of entire cases, respectively, indicating that musculoskeletal disorders are the leading cause of requiring care and support(4). The global burden of disease study conducted by the World Health Organization in 2010 reported that musculoskeletal disorders are the main factor affecting the number of years lived with disability(5). Therefore, the establishment of preventive measures against lifestyle degradation associated with musculoskeletal decline is an urgent issue worldwide.
Changes in body composition associated with aging are known to be deeply involved in lifestyle degradation in older individuals, and decreased skeletal muscle mass (SMM) in particular is associated with physical dysfunction and disabilities (6-9). SMM decreases 0.47% per year in men and 0.37% per year in women, and by the age of 75 years, this decrease rises to 0.80%–0.98% per year in men and 0.64%–0.70% per year in women (10). Thus, decreased SMM associated with aging, also known as sarcopenia, has attracted attention as a likely cause of various future disorders. The European Working Group on Sarcopenia in Older People (EWGSOP) (11) and the Society on Sarcopenia, Cachexia and Wasting Disorders (12) have both introduced diagnostic criteria for sarcopenia. Each criterion includes decreased SMM as an essential criterion, with muscular weakness and decreased physical performance also as included as important criteria for diagnosing sarcopenia. The detection of marked muscular weakness and decreased physical performance is easy if the decrease in SMM has already progressed but is not always sufficient for early detection and prevention. A new index that predicts decreases in SMM earlier was developed to enable early detection and intervention before sarcopenia develops. Most of recent prospective studies on frail older individuals with sarcopenia have examined the course of the disease over a relatively long period of time from the baseline survey (13, 14). However, from the perspective of prevention and early detection, it may also be important to predict SMM decreases over a relatively short period of time. Therefore, the aim of this study was to elucidate SMM decreases and related factors over a relatively shorter period of time.
Methods
Subjects
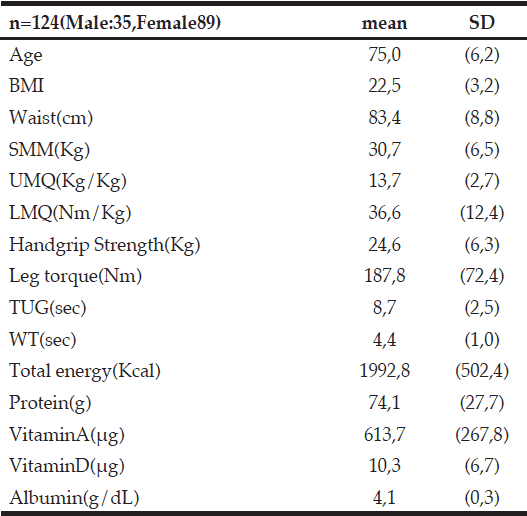
A total of 387 elderly individuals aged 65 years or older who were residents of the city of Yokkaichi, Mie Prefecture, and the city of Nagoya and Handa, Aichi Prefecture, participated in the present study. Of these, 124 participated in continuous data collection in 2010 and 2011 (male = 35, mean age = 75.6 ± 6.1 years; female = 89, mean age = 74.8 ± 6.3 years).
The purpose of the study was explained to all the participants before obtaining written informed consent.
Anthropometric and muscle mass measurements
Height was measured to the nearest 0.1 cm using a stadiometer, and weight was measured to the nearest 0.1 kg (Inbody230, BioSpace Co., Ltd.). To adjust for clothing, the final value was the measured value minus 1 kg. Body Mass Index (BMI) was calculated by dividing weight by height squared. Maximal knee extension strength was measured using a hand-held dynamometer (μTas-1; ANIMA Ltd.) for those who were able to lift the foot independently. During testing, participants sat on a hard chair with the knee and hip joints at 90° of flexion and were strongly encouraged to exhibit the greatest possible force. To ensure the knee joint was at 90°, a belt fitted with a strain gauge-type pressure sensor was placed on the distal portion of the subject’s leg and fixed to the rear legs of the chair. Measurements were performed with the non-dominant leg; the isometric extension strength was measured twice for more than 3 s. The strength was measured as a peak force and expressed as the kilograms of force the examiner had to apply to break the isometric contraction. The best result of the two trials was used in the analyses, unless only one result was available. The dynamometer was placed proximal to the ankle joint. Grip strength was measured using a Smedley Hand Dynamometer with the upper extremity hanging naturally at the side of the body; the proximal interphalangeal joint of the index finger was adjusted to 90°. These measurements were conducted twice for both the left and right sides, and the result of the maximum value (to the nearest 0.1 kg) was used. Walking time (WT) was measured with a stopwatch and recorded accurate to 0.1 s; the test measured the time it took to walk 5 m after a 3-m run-up on a flat surface. A single measurement was taken for walking speed. The Timed Up and Go (TUG) test involved rising from a chair, walking 3 m, turning around, walking back to the chair, and sitting down. The starting posture involved leaning back slightly on the chair’s backrest with the hands placed on the thighs. The TUG test is one of the most frequently used tests of balance and gait and is often used to assess fall risk in older people. The time to complete the TUG test was measured in seconds at each participant’s usual pace. The 0-m point was the front legs of the chair, and the 3-m point was the center of a cone. Researchers measured the time from when the subject’s body began to move until their backside came in contact with the chair again. Subjects were free to go around the cone in their own way, and the smallest (fastest time) of the two measured values was used. Measurements were taken with a stopwatch accurate to 0.1 s. Walking speed was at the subject’s usual speed .
To estimate the energy and nutrient intake of each subject during the previous 1–2 months, all subjects were interviewed by experienced dietitians with the Excel Eiyo-kun (nutrition) Food Frequency Questionnaire based on food groups (FFQg, Ver 2.0) using Japanese food composition tables, which is based on 29 food groups and 10 types of cooking. This questionnaire was developed by Takahashi(15) and is based on Japanese data. Its validity is comparable with dietary record methods, and other reports of dietary surveys have already used this questionnaire(16-17) .
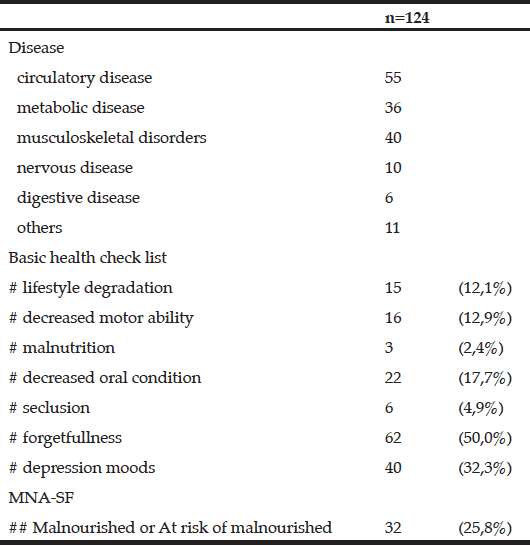
Nutritional states were assessed using albumin levels and the Mini Nutritional Assessment Short Form (MNA-SF). A basic health checklist for those over 65 years old was used to assess lifestyle. In the checklist, any lifestyle degradation, decreased motor ability, malnutrition, decreased oral condition, seclusion, forgetfulness, and emotions were assessed.
Site-specific SMM was measured with an impedance measurement device (Inbody230, BioSpace Co., Ltd.) using two different frequencies (20 Hz and 100 Hz) and a tetrapolar 8-point tactile electrode system. Muscle quality was assessed by calculating the upper extremity muscle quality (UMQ), where right grip strength was divided by right upper extremity muscle mass, and lower extremity muscle quality (LMQ), where right knee extension strength was divided by right lower extremity muscle mass.
Analysis
Study variables were analyzed by calculating the difference between SMM at baseline and SMM 1 year later. Participants were divided into three groups based on SMM difference. The first tertile group, which had the greatest decrease in SMM, was compared with the second/third tertile group. Comparisons at baseline were conducted using the unpaired t-test for quantitative variables and the chi-squared test for nominal variables. To extract the factors related with a significant decrease in SMM, a logistic regression analysis and Classification and Regression Trees (CART) were used for multivariate analysis. SPSS Version 19.0 (IBM Corp, USA) was used for statistical processing, and a p level of < .05 was considered to show statistical significance throughout analyses.
This study was carried out in compliance with the Declaration of Helsinki under the approval of the Bioethics Review Board of the Nagoya University Graduate School of Medicine. The purpose of the study was explained to each participant individually at the time of recruitment, and the individuals gave written consent to participate. Sufficient caution was paid to having an examiner present to prevent the older individuals from falling during examinations.
Results
Tables 1 and 2 show the basic characteristics of subjects. Table 3 shows the comparisons of ages, body composition, SMM, UMQ, LMQ, grip strength, leg torque, TUG, and WT between the first tertile and second/third tertile groups. In the first tertile and second/third tertile groups, significant differences were observed for age (77.7 ± 5.8 years and 73.6 ± 6.0 years, respectively), SMM (31.9 ± 6.4 and 30.4 ± 6.4, respectively), UMQ (12.4 ± 2.6 and 14.1 ± 2.5, respectively), TUG (9.8 ± 2.7 and 8.1 ± 2.3, respectively), and WT (4.8 ± 9.1 and 4.2 ± 0.9, respectively). No significant differences were observed for BMI, LMQ, grip strength, leg torque, or the levels of protein, vitamin A, vitamin D, or albumin. However, a small effect size was observed for SMM (r = 0.11) and vitamin D intake (r = 0.16).
BMI,Body Mass Index; SMM, Skeletal Muscle mass; UMQ,Upper Extremity Muscle Quality; LMQ,Lower Extremity Muscle Quality; TUG,Time up and Go; WT, 5m walking Time; Total energy, Protein,VitaminA,VitaminD culculate from FFQg
Disease allow multiple answers; MNA-SF, mini nutritional assessment short-form; # Calculated from the basic checklist for those over 65 years old; ## Calculated from MNA-SF
Table 4 shows the results for gender, the basic checklist, and the MNA-SF at baseline for the first tertile and second/third tertile groups. No significant differences were observed for any of the items. A small effect size was observed for gender (Φ = 0.16), seclusion (Φ = 0.16), forgetfulness (Φ = 0.11), drop in mood (Φ = 0.13), and the MNA-SF (Φ = 0.15).
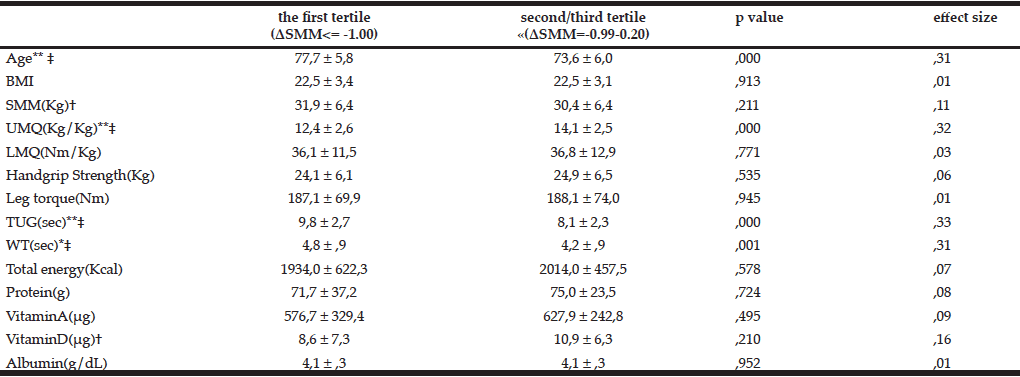
Table 3 Comparison of the amount of SMM change at baseline between the first tertile and second/third tertile groups (scale)
* p≤0.05 **p≤00.01; t-test; † small effect size, 0.1≤effect size<0.3; ‡ medium effect size, 0.3≤effect size<0.5; WT, 5m walking Time; Total energy, Protein,VitaminA,VitaminD culculate from FFQg
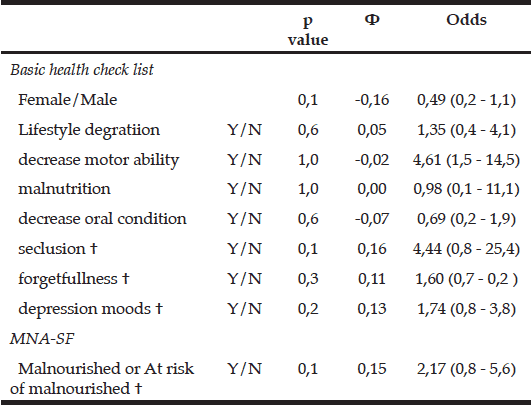
Table 4 Comparison of the amount of SMM change at baseline between the first tertile and second/third tertile groups (categories)
chi-squared test; † small effect size, 0.1≤Φ<0.3
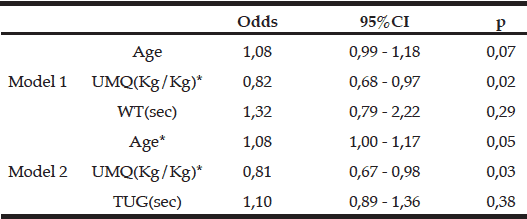
Table 5 shows correlations between variables. Because significantly higher correlation was found between WT and TUG as expected (r = 0.79), these variables were not input simultaneously in the logistic regression analysis due to predicted collinearity. Table 6 shows the results of the logistic regression analysis. UMQ (odds = 0.82, 0.81) was chosen as a significant variable in both models, and age (odds = 1.08) was chosen as a significant variable in model 2.
* p<0.05; ** p<0.01; UMQ,Upper Extremity Muscle Quality; TUG,Time up and Go; WT, 5m walking Time
Dependent variable (1= first tertile, 0 = second/third tertile); Independent variable ; age, SMM, UMQ, VitaminD, seclusion, forgetfulness, depression moods,WT or TUG; * p≤0.05; UMQ, Upper Extremity Muscle Quality; WT, 5m walking Time; TUG, Time Up and Go
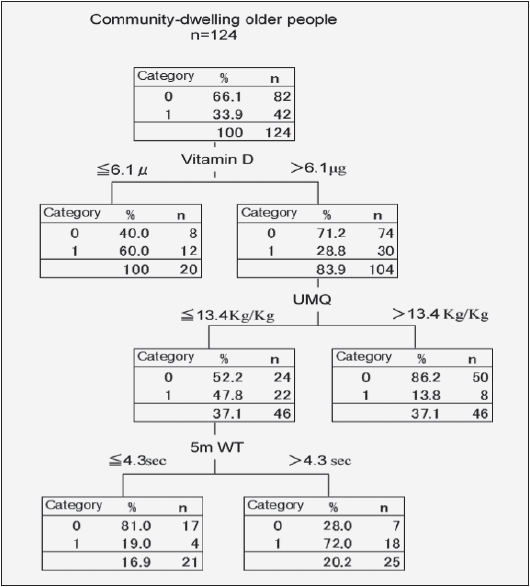
Figure 1 shows the CART decision tree. The percentage of correct answers among subjects was 78.2% (95% confidence interval, 77.4%–79.0%), which were classified into four terminal nodes. Vitamin D, UMQ, and WT were chosen as factors for a significant decrease in SMM. In the present model, vitamin D was the first choice in the first tier, and 6.05 g was the limit where the results branched into two groups. In the group where vitamin D intake exceeded 6.05 g, the second tier results branched depending on the UMQ; at the third tier, results branched depending on 5-m walking time (4.33 s).
Category 1 is the first tertile and Category 0 is the second/third tertile; Dependent variable (1= first tertile, 0 = second/third tertile); Independent variables; age, SMM, UMQ, VitaminD, seclusion, forgetfulness, depression moods,WT, TUG; UMQ, Upper Extremity Muscle Quality; WT, 5m Walking Time
Discussion
Decreased SMM as a result of aging is unavoidable and is associated with physical dysfunction and disabilities (9). In our comparison of the first tertile, which had the greatest decrease in SMM, and the second/third tertile groups, significant differences were observed for age, UMQ, TUG, and 5-m WT. A small effect size was observed for SMM and vitamin D. Although no significant differences were observed for items from the basic checklist assessing daily functional status, a small effect size was observed for seclusion, dementia, and emotions. The results are in keeping with a report by Baumgartner et al(18) showing that in males, decreased SMM associated with aging was related to the occurrence of falls in the past year, the use of a walker or cane, decreased balance function, and instrumental ADL limitations, while in females it was related to instrumental ADL limitations. Factors related to these findings include gender, age, menopause, height, weight, BMI, amount of body fat, physical activity, carotenoids, vitamin D, amino acid branched chains, and protein intake, all of which are known to be multiple risk factors (19-21). In the present study, no significant difference was observed for instrumental ADLs in community-dwelling older individuals leading independent lives. However, symptoms of seclusion, forgetfulness, and depression may have appeared prior to issues with instrumental ADLs. According to Fried (22), older individuals gradually become weaker in a cyclic fashion: the total amount of energy is reduced due to a decrease in physical activity and decreased appetite leads to malnutrition, which in turn leads to decreased SMM. The consequent reduction in muscle strength and aerobic exercise capacity lowers walking function and results in limited activity. Furthermore, decreased SMM lowers basal metabolism and accelerates the decrease in total energy. Seclusion, forgetfulness, and depression are linked to reduced physical activity and may be the cause of decreased SMM.
Many previous studies investigating the relevant factors associated with decline in SMM and motor ability have used logistic regression analyses and multiple regression analyses (9-13). These analyses are useful indicators for assessing the effect of independent factors and excluding the effect of other factors. However, these regression analyses have the following drawbacks: (1) the assumption of a linear relationship between dependent variables and explanatory variables, (2) chosen variables can be influenced by the presence of multicollinearity, (3) complicated prediction formulas making clinical application difficult, and (4) data with missing values being useless in the analyses. Decision tree analyses align factors hierarchically in order from the factor most strongly related to the dependent variable; thus, the relationship between each factor is easy to interpret. Moreover, unlike logistic regression or multiple regression analysis, multicollinearity between variables theoretically has no influence on the results in the present analysis (23, 24). Decreased muscle strength, decreased SMM, and physical dysfunction associated with aging have a nonlinear relationship (25, 26); therefore, it is valid to use a decision tree analysis. In the present decision tree analysis, vitamin D, UMQ, and WTwere extracted as factors related with decreased SMM. According to a study on muscle tissue collected during a series of surgeries for proximal femoral fractures, type II muscle fibers, which are often found in fast muscle, had atrophied in the vitamin D-deficient group as compared with the vitamin D-sufficient group; the diameter of the type II fibers was significantly correlated with serum vitamin D levels (27). Given the identified distribution of vitamin D receptors in skeletal muscles, vitamin D deficiency can be a factor of decreased muscle mass and strength. Selective atrophy of type II fibers with large muscle output, expansion of small diameter type I fibers (28), and a decrease in the number of motor units are observed in skeletal muscle as a result of aging (20). During the process of aging, interactions between muscles and motor neurons via neuromuscular synapses can be attenuated, thus leading to muscular weakness (29). The quality of muscle, assessed by muscle strength per unit weight, is believed to decline as a result of these factors. In addition to muscle strength and SMM, the quality of muscle deserves mention as an important variable for predicting decreased SMM. A previous study comparing the relationship between walking and SMM reported that decreased SMM was associated with walking function (30). Because factors in the decision tree are chosen in the order from the factor most strongly affected by the dependent variable, walking function is the next affected factor after vitamin D intake and UMQ. When a decrease was observed in UMQ, decreased walking function was found to increase the risk of decreased SMM.
The variables extracted as factors related to decreased SMM in the present study were chosen from items in a previous study(20) therefore considered valid. By adopting a decision tree in the present analysis, we were able to visually comprehend the extent of involvement and correlation between decreased SMM and each variable. We believe that the present index is appropriate for practical use in clinical settings. However, as this study was an observational study, older individuals who maintained relatively higher awareness about one’s own health served as samples, leading to unavoidable selection bias. Moreover, activities such as work and hobbies and social circumstances such as family structure were not included in the study items. While estimates for daily vitamin D intake being taken from the food frequency questionnaire, actual serum vitamin D levels were not measured.
Conclusion
The results indicated that vitamin D intake, UMQ, and WT are related with decreased SMM in community-dwelling older individuals. As aforementioned, decreased SMM associated with aging is unavoidable. However, we believe that the present study provided an evidence suggesting individuals at risk of developing decreased SMM can be screened by simple anthropometric or clinical surrogates before they progress to sarcopenia .Thus establishing validity of such surrogate markers may contribute to increasing the disability-adjusted life expectancy by early interventional approaches.
Conflicts of Interest: The authors declare no financial conflicts of interest.
References
1. Ministry of Health, Labour and Welfare. Annual Report on Health, Labor and Welfare, 2010-2011.(Tokyo: Nikkei PRINTING INC) 2011.
2. Topinkova, E. Aging, disability and frailty. Annals of nutrition & metabolism 2008;52 Suppl 1, 6-11.
3. Minoru, y. Managing sarcopenia with nutrition and exercise in older adults. J. JSPEN 2013;28, 1065-1068.
4. Statistics and Information Department, Ministry of Health, Labour and Welfare. Comprehensive Survey of Living Conditions.(Tokyo: Health and Welfare Stastics Association) 2007.
5. Salomon, J.A., Wang, H., Freeman, M.K., Vos, T., Flaxman, A.D., Lopez, A.D., and Murray, C.J.L. Healthy life expectancy for 187 countries, 1990–2010: a systematic analysis for the Global Burden Disease Study 2010. The Lancet 2012;380, 2144-2162.
6. Nishiwaki, T., Nakamura, K., Ueno, K., Fujino, K., and Yamamoto, M. Health characteristics of elderly Japanese requiring care at home. The Tohoku journal of experimental medicine 2005;205, 231-239.
7. Janssen, I. ( Influence of Sarcopenia on the Development of Physical Disability: The Cardiovascular Health Study. Journal of the American Geriatrics Society 2006;54, 56-62.
8. Volpato, S., Romagnoni, F., Soattin, L., Ble, A., Leoci, V., Bollini, C., Fellin, R., and Zuliani, G. Body mass index, body cell mass, and 4-year all-cause mortality risk in older nursing home residents. J Am Geriatr Soc 2004;52, 886-891.
9. Janssen, I., Heymsfield, S.B., and Ross, R. Low Relative Skeletal Muscle Mass (Sarcopenia) in Older Persons Is Associated with Functional Impairment and Physical Disability. Journal of the American Geriatrics Society 2002;50, 889-896.
10. Mitchell, W.K., Atherton, P.J., Williams, J., Larvin, M., Lund, J.N., and Narici, M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. . Front Physiol 2012;260.
11. Cruz-Jentoft, A.J., Baeyens, J.P., Bauer, J.M., Boirie, Y., Cederholm, T., Landi, F., Martin, F.C., Michel, J.-P., Rolland, Y., Schneider, S.M., et al. Sarcopenia: European consensus on definition and diagnosi. Age Ageing 2010;39, 412-423.
12. Morley, J.E., Abbatecola, A.M., Argiles, J.M., Baracos, V., Bauer, J., Bhasin, S., Cederholm, T., Stewart Coats, A.J., Cummings, S.R., Evans, W.J., et al. Sarcopenia With Limited Mobility: An International Consensus. Journal of the American Medical Directors Association 2011;12, 403-409.
13. Koster, A., Ding, J., Stenholm, S., Caserotti, P., Houston, D.K., Nicklas, B.J., You, T., Lee, J.S., Visser, M., Newman, A.B., et al. Does the Amount of Fat Mass Predict Age-Related Loss of Lean Mass, Muscle Strength, and Muscle Quality in Older Adults? The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2011;66A, 888-895.
14. Auyeung, T.W., Lee, J.S., Leung, J., Kwok, T., and Woo, J. Adiposity to muscle ratio predicts incident physical limitation in a cohort of 3,153 older adults-an alternative measurement of sarcopenia and sarcopenic obesity. Age (Dordr), 2012.
15. Talahasi, K. Food Frequency Questionnaire Based on Food Groups for Estimating Individual Nutrient Intake. TheJapaneseJournalofNutritionandDietetics 2003;61, 161-169.
16. Masuda, R., Imamura, H., Mizuuchi, K., Miyahara, K., Kumagai, H., and Hirakata, H. Physical activity, high-density lipoprotein cholesterol subfractions and lecithin:cholesterol acyltransferase in dialysis patients. Nephron Clinical practice 2009;111, c253-259.
17. Kawamata, k., Obuchi, S., and Tomotake, H. The Effects of Comprehensive Geriatric Training on Physical Performance, Dietary Intake, and Blood Composition for Local Elderly People −A Case in The Southern Nagano Region of Japan−. Jpn J Phys Fitness Sports Med 2012;61, 495-502.
18. Baumgartner, R.N., Koehler, K.M., Gallagher, D., Romero, L., Heymsfield, S.B., Ross, R.R., Garry, P.J., and Lindeman, R.D. Epidemiology of Sarcopenia among the Elderly in New Mexico. American Journal of Epidemiology 1998;147, 755-763.
19. Landi, F., Liperoti, R., Fusco, D., Mastropaolo, S., Quattrociocchi, D., Proia, A., Russo, A., Bernabei, R., and Onder, G. Prevalence and Risk Factors of Sarcopenia Among Nursing Home Older Residents. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2012;67A, 48-55.
20. Doherty, T.J. Invited Review: Aging and sarcopenia. Journal of Applied Physiology 2003;95, 1717-1727.
21. Shimokata, H., and Ando, F. Sarcopenia and its risk factors in epidemiological study. Nihon Ronen Igakkai zasshi Japanese journal of geriatrics 2012;49, 721-725.
22. Fried, L.P., Tangen, C.M., Walston, J., Newman, A.B., Hirsch, C., Gottdiener, J., Seeman, T., Tracy, R., Kop, W.J., Burke, G., et al. Frailty in Older Adults : Evidence for a Phenotype. the journal of gerontology series a 56, M146-M156. 2001
23. Kim, J., Tanabe, K., Yokoyama, N., Zempo, H., and Kuno, S. Association between physical activity and metabolic syndrome in middle-aged Japanese: a cross-sectional study. BMC Public Health 2011;11, 624.
24. Arai, T., Kaneko, S., and Fujita, H. Decision trees on gait independence in patients with femoral neck fracture. Nippon Ronen Igakkai Zasshi 2011;48, 539-544.
25. Goodpaster, B.H., Park, S.W., Harris, T.B., Kritchevsky, S.B., Nevitt, M., Schwartz, A.V., Simonsick, E.M., Tylavsky, F.A., Visser, M., and Newman, A.B. The Loss of Skeletal Muscle Strength, Mass, and Quality in Older Adults: The Health, Aging and Body Composition Study. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2006;61, 1059-1064.
26. Rantanen, T., Guralnik, J.M., Ferrucci, L., Penninx, B.W.J.H., Leveille, S., Sipilä, S., and Fried, L.P. Coimpairments as Predictors of Severe Walking Disability in Older Women. Journal of the American Geriatrics Society 2001;49, 21-27.
27. Sato, Y., Inose, M., Higuchi, I., Higuchi, F., and Kondo, I. Changes in the supporting muscles of the fractured hip in elderly women. Bone 2002;30, 325-330.
28. Nikolic, M., Malnar-Dragojevic, D., Bobinac, D., Bajek, S., Jerkovic, R., and Soic-Vranic, T. Age-related skeletal muscle atrophy in humans: an immunohistochemical and morphometric study. Collegium antropologicum 2001;25, 545-553.
29. Bütikofer, L., Zurlinden, A., Bolliger, M.F., Kunz, B., and Sonderegger, P. Destabilization of the neuromuscular junction by proteolytic cleavage of agrin results in precocious sarcopenia. The FASEB Journal 2011;25, 4378-4393.
30. Kim, J., Kuno, S., Soma, R., Masuda, K., Adachi, K., Nishijima, T., Ishizu, M., and Okada, M. Relationship between reduction of hip joint and thigh muscle and walking ability in elderly people. The Japanese Journal of Physical Fitness and Sports Medicine 2000;49, 589-596.