S. Lindskov1,2,3, K. Sjöberg4, A. Westergren1, P. Hagell1
1. The PRO-CARE group, School of Health and Society, Kristianstad University, Kristianstad, Sweden; 2. Department of Geriatrics and Neurology, Central Hospital Kristianstad, Northeast Skåne Health Care District, Kristianstad, Sweden; 3. Department of Clinical Sciences, Lund University, Lund, Sweden; 4. Department of Clinical Sciences, Lund University, Division of Gastroenterology and Nutrition, Skåne University Hospital, Malmö, Sweden.
Corresponding Author: Susanne Lindskov, School of Health and Society, Kristianstad University, SE-291 88 Kristianstad, Sweden, Tel: +46 44 20 85 63, E-mail: Susanne.Lindskov@hkr.se
Abstract
Background: Unintentional weight loss and undernutrition have been found common in Parkinson’s disease but its relation to other disease aspects is unclear. Objectives: To explore nutritional status in relation to disease duration in Parkinson’s disease, as well as associations between nutritional status and motor and autonomic features. Design: Cross-sectional. Setting: South-Swedish outpatient Parkinson-clinic. Participants: Home-dwelling people with Parkinson’s disease (n=71), without significant cognitive impairment (mean age, 67.3 years; 56% men; mean disease duration, 6.3 years). Measurements: Parkinsonian motor symptoms, mobility, activity level, disability, dyskinesias, dysautonomia, under- and malnutrition risk screening (using MEONF II and MUST for undernutrition and SCREEN II for malnutrition) and anthropometric measures (BMI, handgrip strength, triceps skin-fold, mid-arm circumference and mid-upper arm muscle circumference) were recorded. The sample was divided into those with longer (n=34) and shorter disease duration (n=37) according to the median (5 years). Results: Longer disease duration was associated with more, disability, dyskinesias and dysautonomia than shorter duration (P≤0.04). Mean (SD) body weight and BMI were 80.3 (16.3) kg and 28.1 (4.8) kg/m2, respectively, and did not differ between duration groups (body weight, 80.9 vs. 79.6 kg; BMI, 28.0 vs. 28.3 kg/m2; P≥0.738). There were no differences in other anthropometric measures between duration groups (P≥0.300). BMI identified 4% and 62% as under- and overweight, respectively, and 4% exhibited undernutrition risk, whereas 87% were at risk for malnutrition. Nutritional and motor/dysautonomic variables showed relatively weak correlations (rs, ≤ 0.33), but people with orthostatic hypotension had lower BMI (26.7 vs 29.2 kg/m2; P=0.026) and lower handgrip strength (33.2 vs 41.6 kg; P=0.025) than those without orthostatic hypotension. Conclusion: Motor and autonomic features showed expected relationships with disease duration. In contrast to these observations, and to most previous reports on nutrition in PD, frequencies of underweight and undernutrition were low. However, malnutrition risk was high, emphasizing the need for regular clinical monitoring of nutritional status. The reasons for the preserved nutritional status have to be explored prospectively.
Key words: Duration, nutrition, Parkinson’s disease, weight.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by motor symptoms such as bradykinesia, rigidity and tremor. Complications such as a fluctuating drug response and dyskinesias often develop over time. Non-motor symptoms, e.g., dysautonomia, are also common (1). One poorly understood feature is unintentional weight loss and undernutrition (2, 3).
Unintentional weight loss has been reported to occur among up to a third of people
with PD (4), and the prevalence of undernutrition and undernutrition risk have been reported to be up to 24% and 60%, respectively (5). A recent meta-analysis of studies reporting body mass index (BMI) among people with PD and healthy controls found a significant difference with an average BMI in PD 1.73 units below that of controls (95% CI, 1.11- 2.35) (6). Although weight loss has been found associated with PD severity rather than duration (6), some studies have found a relationship with disease duration (7), and it may also occur early in the disease, even before PD onset despite an increased energy intake (8). Furthermore, longitudinal data have suggested an association with PD duration with decreasing BMI and increasing risk for undernutrition over time (9).
However, a weakness of many previous studies in this area is that most of them have only examined BMI or unintentional weight loss, without addressing nutritional status in a broader sense by the use of valid and reliable tools and indicators for nutritional status.
The cause of weight loss and undernutrition in PD remains unknown, but reduced energy intake and/or increased energy expenditure have been suggested as possible factors (4). However, there is also evidence against this explanation (10), and other factors may also contribute. For example, since autonomic centers such as the hypothalamus are involved in weight control, pathology within such areas may be related to weight loss (10). Furthermore, insufficient awareness of nutritional risks and failure to monitor nutritional status may also contribute.
The objectives of this study were to explore nutritional status, motor and autonomic features in relation to disease duration in PD, as well as the association between nutritional status and motor and autonomic features.
Methods
The study setting was a multidisciplinary outpatient PD clinic at a South-Swedish central hospital serving a population of about 170 000. Ninety-eight consecutive people with idiopathic PD were invited to participate. Inclusion criteria were independent living and absence of clinically significant cognitive impairment (as determined by the attending clinician and routine cognitive screening (11)). All participants provided written informed consent. The study was approved by the regional Research Ethics Committee.
Procedures and data collection
The week before the study visit, participants were sent a booklet of patient-reported rating scales to be completed before the clinic visit. All visits were scheduled in the morning at about 10-11 am following a light breakfast. All data collection was conducted by the same assessor, a PD specialized nurse trained in using the rating scales, nutritional screening tools and anthropometric measures employed here.
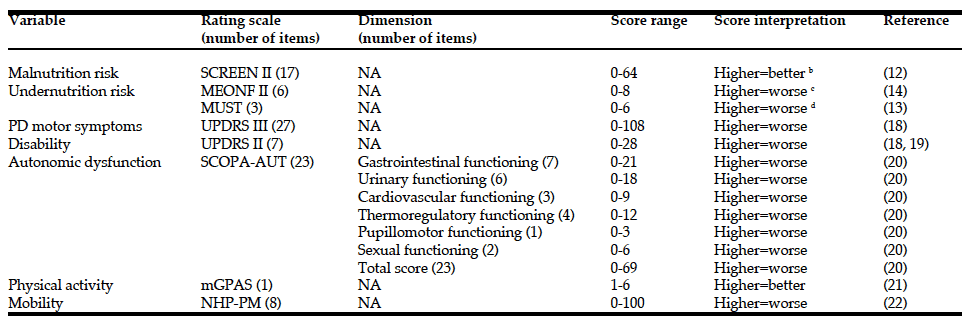
Nutritional status was screened by using the SCREEN II (12), MUST (13) and MEONF II (14) (Table 1). MEONF II and MUST are clinical undernutrition screening tools, whereas SCREEN II is a tool for screening of malnutrition in general (not just undernutrition). Although the MEONF II has been found to display advantages compared to the MUST (13) we used both because the MUST is more widely known to have previous PD studies (7). Anthropometric measures (15, 16) included body weight (kg), BMI (weight in kg/height in meters2), estimation of body muscle and fat mass by Triceps Skin Fold (TSF; mm) and Mid Arm Circumference (MAC; cm). Mid-Upper Arm Muscle Circumference (MUAMC) was calculating based on TSF and MAC using the formula: MUAMC (cm) = MAC – 0.1 x TSF). In addition, Hand Grip Strength (HGS; kg) was measured in the right hand. Body weight and height were measured using standard clinical equipment, an analog scale (Stathmos-Lindell, Sweden) and a stadiometer (Hultafors, Sweden), respectively, with patients wearing light clothing and no shoes. TSF was measured with a caliper (Skinfold Caliper Baseline, Enterprises Inc., USA) at the back of the upper arm. Subcutaneous fat was gripped 1 cm above the midpoint between the shoulder (acromion) and the tip of the elbow when the arm was hanging and relaxed. MAC was measured using a flexible measuring tape (included with the TSF caliper), halfway between the shoulder (acromion) and the tip of the elbow. HGS was measured using the Baseline Hydraulic Hand Dynamometer (Enterprises Inc., USA), with a capacity of 90 kg.
a. All scales are patient-reported except for the MEONF II, UPDRS II (clinical interview and observation) and UPDRS III (clinical examination); b. Risk cut-off scores: <54 = any risk; <50 = high risk; c. Risk cut-off scores: >2 = moderate risk; >4 = high risk; d. Risk cut-off scores: 0 = low risk; 1 = medium risk; ≥2 = high risk; SCOPA-AUT, SCales for Outcomes in PArkinson’s disease – Autonomic symptoms; mGPAS, modified Grimby Physical Activity Scale; SCREEN II, Seniors in the Community: Risk Evaluation for Eating and Nutrition, version II; MEONF II, Minimal Eating Observation and Nutrition Form – version II; MUST, Malnutrition Universal Screening Tool; NHP-PM, Physical Mobility section of the Nottingham Health Profile; UPDRS III, Unified Parkinson’s Disease Rating Scale, part III (motor examination); UPDRS II, Unified Parkinson’s Disease Rating Scale, part II (activities of daily living); NA, not applicable
Anthoprometric measures were recorded as the mean of three consecutive measurements, and cut-off scores were applied as recommended in the literature, including age- adjusted BMI classifications (≤69 years old, BMI ˂20 = underweight; BMI ≥25 = overweight/obese, ≥70 years old, BMI ˂22 = underweight; ≥27 = overweight/obese) (15-17).
The presence or absence of dyskinesias was noted, and PD motor symptoms were assessed by part III (motor examination) of the Unified PD Rating Scale (UPDRS III) (18) during the “on” phase (i.e., periods with good antiparkinsonian drug response). Disability was assessed by the disability score (19) of UPDRS part II (activities of daily living) both for the “on” and the “off” (periods with poor drug response and increased disability) phases. The UPDRS III includes rating of the presence and severity of various motor symptoms while conducting a standardized neurological examination and the UPDRS II disability score includes ratings of various patient- reported disabilities in daily life as assessed during a standardized interview (18). Autonomic dysfunction, physical activity and mobility were assessed by patient- reported rating scales (the SCales for Outcomes in PArkinson’s disease – Autonomic symptoms (SCOPA- AUT), the modified Grimby Physical activity scale (mGPAS), and the Physical Mobility section of the Nottingham Health Profile (NHP-PM), respectively). Further details on all applied rating scales and screening tools (12-14,18-22) are summarized in Table 1. In addition, orthostatic hypotension (OH) was determined by blood pressure measurements using a manual blood pressure cuff (Jewel Movement Sphygmomanometer, AB Henry Eriksson, Sweden) after 10 minutes rest, immediately after standing up, and following 3 minutes of standing (23).
Analyses
Data were checked regarding underlying assumptions and described and analyzed accordingly using IBM SPSS 20 (Armonk, NY: IBM Corp.) and Confidence Interval Analysis 2.2 (www.som.soton.ac.uk/cia/). The alpha- level of significance was set at 0.05 (2-tailed). We did not adjust for multiple testing due to the exploratory nature of the study. The sample was divided into those with shorter (˂5 years) and longer (≥5 years) PD duration according to the median, and variables were compared between these groups using chi-squared, Mann-Whitney and independent samples t-tests, as appropriate; 95% confidence intervals (CIs) were calculated. Spearman correlations were computed between disease duration and nutritional variables, and between nutritional variables and motor and autonomic scores. Nutritional variables were also compared between those with and without orthostatic hypotension (Mann-Whitney and independent samples t-test, as appropriate).
Results
Twenty seven (28%; 16 women; mean (min-max) age and PD duration, 71 (60-87) and 4.8 (1-15) years, respectively) of the 98 invited patients did not respond to the study invitation. The final sample consisted of 71 participants (40 men) with a mean (SD; min-max) age of (8.1; 47-89) years who had been diagnosed with PD for a mean (SD; min-max) of 6.3 (3.6; 0.5-18) and median (q1-q3) of 5 (4-8) years. Fifty-three participants (74%) were married/living as married, and the majority (65%) was retired while the rest were either working (28%) or on long-term sick leave/disability retirement (7%). About two thirds (68%) had some comorbidity. Pharmacological PD treatment consisted of levodopa (n=70), dopamine agonists (n=63), COMT-inhibitors (n=45), MAO-B- inhibitors (n=11), and amantadine (n=3). Two participants had undergone thalamic deep brain stimulation, and one was not on any medical antiparkinsonian therapy.
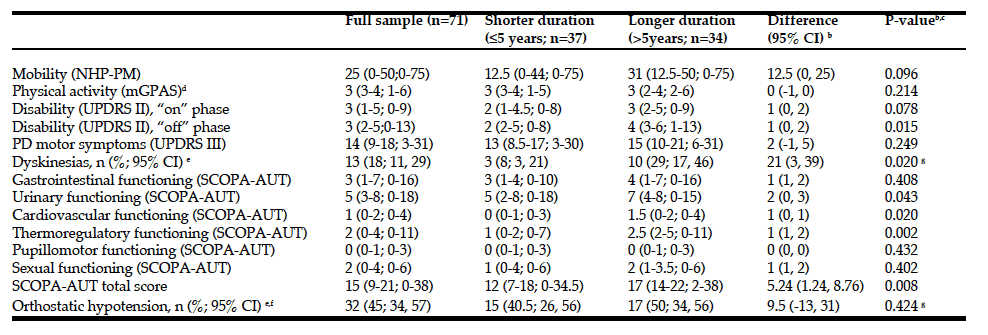
a. Data are median (q1-q3; min-max) unless otherwise noted; higher scores = worse unless otherwise noted; b. Differences in percentages (dyskinesias and orthostatic hypotension) and medians (all other variables) between people with longer (˃5 years) vs shorter (≤5 years) PD duration; c. Mann-Whitney tests (unless otherwise noted); d. Higher scores = better; e. 95% confidence intervals for percentages; f. Orthostatic hypotension was defined as a decrease in systolic/diastolic blood pressure of ≥20/10 mmHg (≥30/15 mmHg in people with hypertension) within 3 minutes of standing (23); g. Chi-squared test; NHP-PM, Physical Mobility section of the Nottingham Health Profile; mGPAS, modified Grimby Physical Activity Scale; UPDRS II, Unified Parkinson’s Disease Rating Scale, part II (activities of daily living); UPDRS III, Unified Parkinson’s Disease Rating Scale, part III (motor examination); SCOPA-AUT, SCales for Outcomes in PArkinson’s disease – Autonomic symptoms.
Motor and autonomic variables are reported in Table 2. PD, disability and dyskinesias, as well as autonomic symptoms (total as well as urinary, cardiovascular and thermoregulatory SCOPA-AUT scores) were more pronounced in the longer duration group (Table 2).
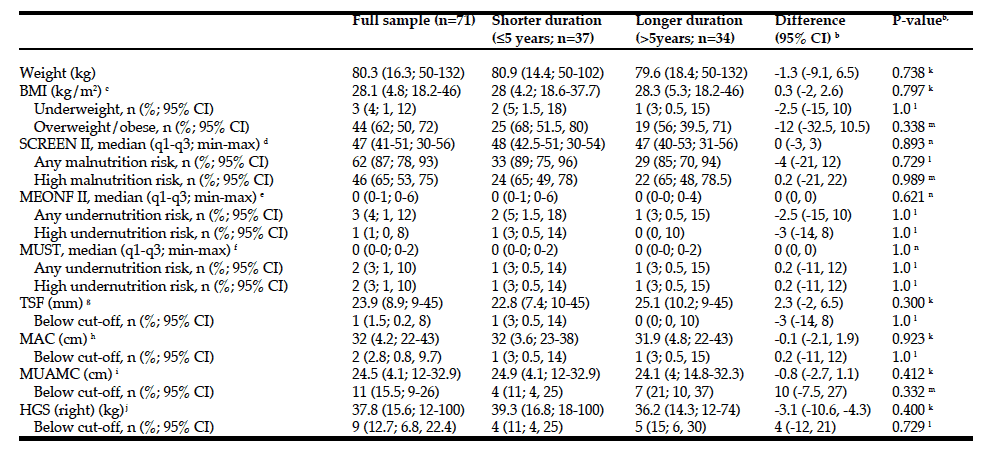
Nutritional data are reported in Table 3. Overall, there were no differences between the two duration groups regarding any nutritional variables. Correlations between disease duration and nutritional variables were non- significant and ranged from -0.01 (MEONF II) to 0.11 (TSF) (Table 4). According to BMI, 3 people (2 shorter and 1 longer PD duration) were underweight and 44 (62%) were overweight (68% in the shorter vs. 56% in the longer duration group). However, 46 participants (65%) exhibited high risk for malnutrition according to the
SCREEN II (same proportion for both duration groups), whereas only 2 (3%) and 3 (4%) were found to have undernutrition risk accordingly to the MUST and MEONF II, respectively (Table 3). BMI indicated underweight for both cases with undernutrition risk according to MUST and for two of those with undernutrition risk according to MEONF II.
Correlations between nutritional variables and motor and autonomic scores (Table 4) showed significant but generally weak associations between BMI and NHP-PM; SCREEN II and SCOPA-AUT/thermoregulatory functioning; MEONF II and SCOPA- AUT/gastrointestinal functioning; MUST and SCOPA- AUT/urinary functioning and SCOPA- AUT/thermoregulatory functioning; TSF and SCOPA- AUT/pupillomotor functioning; and between HGS and NHP-PM, UPDRS II/”on”-phase disability, SCOPA- AUT/thermoregulatory functioning, and SCOPA- AUT/pupillomotor functioning. Other correlations were weaker and non-significant (Table 4).
People with OH (n=32) had lower BMI (mean (SD), 26.7 (4.1) vs 29.2 (5.0) kg/m2, respectively; P=0.026) and also lower HGS (mean (SD), 33.2 (11.1) vs 41.6 (17.8) kg, respectively; P=0.025) than those without OH. There were no differences between these groups on any of the other nutritional variables (weight, SCREEN II, MEONF II, MUST, TSF, MAC, MUAMC; data not shown).
a. Data are mean (SD; min-max) unless otherwise noted; b. For differences in percentages (BMI; mal- and undernutrition classifications; TSF, MAC, MUAMC and HGS according to cut-offs), medians (SCREEN II and MEONF II scores) and means (all other variables) between people with longer (˃5 years) vs shorter (≤5 years) PD duration; c. BMI cut-off scores: ≤69 years old, BMI ˂20 = underweight; ≥70 years old, BMI ˂22 = underweight; ≤69 years old, BMI ≥25 = overweight/obese; ≥70 years old, BMI ≥27 = overweight/obese (17); d. Risk cut-off scores: <54 = any risk; <50 http://abilifygeneric-online.com/catalog/Depression/Paxil.htm = high risk (12); e. Risk cut-off scores: >2 = moderate risk; >4 = high risk (14); f. Risk cut-off scores: 0 = low risk; 1 = medium risk; ≥2 = high risk (13); g. Cut-off scores: Men, ≤6; Women, ≤12 (15); h. Cut-off scores: Men ≤79 years old, ≤26; Men >79 years old, ≤24; Women, ≤79 years old, ≤24; Women >79 years old, ≤22 (15); i. Cut-off scores: Men ≤79 years old, ≤23; Men >79 years old, ≤21; Women, ≤79 years old, ≤19; Women >79 years old, ≤18 (15); j. Cut-off scores: Men, <30; Women, <20 (15, 16); k. Independent samples t-test; l. Fisher’s exact
test; m. Chi-squared test; n. Mann-Whitney test; BMI, body mass index; SCREEN II, Seniors in the Community: Risk Evaluation for Eating and Nutrition, version II; MEONF II, Minimal Eating Observation and Nutrition Form – version II; MUST, Malnutrition Universal Screening Tool; TSF, Triceps Skin Fold; MAC, Mid Arm Circumference; MUAMC, Mid-Upper Arm Muscle Circumference; HGS, Hand Grip Strength.
Discussion
Although undernutrition and low BMI have been frequently reported in PD, related to disease severity and (to a lesser extent) duration (5, 6, 10), we found no evidence for prevalent underweight or undernutrition risk. Motor and autonomic symptoms differed by PD duration as expected. However, associations between nutritional status and disease duration and severity were absent or weak, but there was an association between OH and BMI and HGS. Since malnutrition does not only include undernutrition, but also overweight/obesity and nutrient deficiencies this was also considered by applying SCREEN II, which in contrast to undernutrition screening identified a majority of participants as at risk for malnutrition. Indeed, a larger proportion of participants were overweight rather than underweight. However, similarly to other nutritional and anthropometric variables there were no or only weak associations between SCREEN II and PD duration and severity.
According to Sheard et al. (3) BMI should be interpreted with caution due to limited sensitivity in identifying undernourishment, and additional methods should therefore also be considered. Indeed, while frequencies were low we also found BMI to be less sensitive in identifying undernutrition than clinical screening using the MEONF II, but equal to that of MUST. This is in agreement with previous data (14).
Our observations contrast to most previous studies. For example, a recent study among Australian community-dwelling people with PD (2, 3) identified 15% as moderately undernourished (none as severely undernourished), despite apparent lack of significant cognitive impairments and similar age, gender distribution, disease duration and autonomic symptom severity as in our sample. Similarly, Jaafar et al. identified 23.5% of their UK sample of community-dwelling people with PD as at risk for undernutrition according to the MUST, and both these studies reported generally lower values of BMI and anthropometric measures than found here (7). Despite other sample similarities, motor symptoms appear to have been well controlled in our cohort as indicated by motor and disability scores. Since underweight and undernutrition in PD has been associated with markers of disease severity (3, 6), this could contribute to our observations. However, while undernutrition risk has been found to increase over time (9), studies have observed that weight loss can occur at any stage, even before PD onset (8, 10). Furthermore, the association between motor symptom severity and unintentional weight loss has been generally weak and inconsistent (5). It therefore appears unlikely that better motor symptom control per se would be a major explanation for our observations. One possiblility is that unintentional weight loss does occur without causing underweight because of a relatively high baseline weight. Such a mechanism was hypothesized to underpin recent observations of prevalent overweight/obesity in a Mexican PD accutane how long work sample (24). Longitudinal observations will be required to address this possibility. Interestingly however, and in line with our observations, a recent 3- year study among people with early PD reported weight gain and increased fat mass (25).
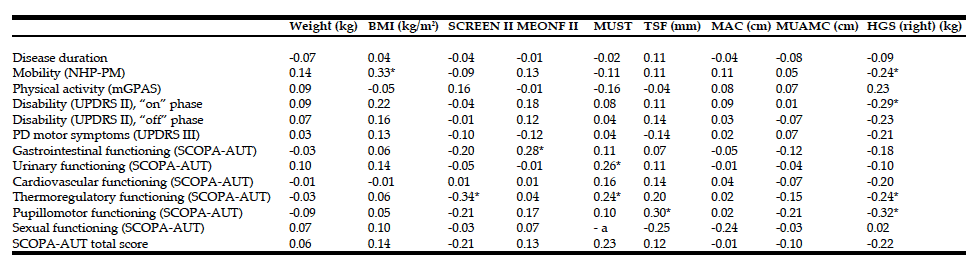
Table 4: Spearman correlations between disease duration and nutritional variables, and between nutritional variables and motor and autonomic scores.
a. Not computable due to constant MUST scores (0) among those with valid Sexual functioning (SCOPA-AUT) scores; * P<0.05. BMI, body mass index; SCREEN II, Seniors in the Community: Risk Evaluation for Eating and Nutrition, version II; MEONF II, Minimal Eating Observation and Nutrition Form – version II; MUST, Malnutrition Universal Screening Tool; TSF, Triceps Skin Fold; MAC, Mid Arm Circumference; MUAMC, Mid-Upper Arm Muscle Circumference; HGS, Hand Grip Strength; NHP-PM, Physical Mobility section of the Nottingham Health Profile; mGPAS, modified Grimby Physical Activity Scale; UPDRS II, Unified Parkinson’s Disease Rating Scale, part II (activities of daily living); UPDRS III, Unified Parkinson’s Disease Rating Scale, part III (motor examination); SCOPA-AUT, SCales for Outcomes in PArkinson’s disease – Autonomic symptoms.
There were few associations between nutritional and autonomic variables. This is in agreement with observations by Sheard et al. (2), who also used the SCOPA-AUT in a community-dwelling sample of people with PD and found a somewhat higher degree of gastrointestinal dysfunction among participants identified as at risk for undernutrition, but no other SCOPA-AUT scores were related to undernutrition. In our study, we also included OH as a more objective autonomic marker and found lower BMI and HGS among people with OH. This is in agreement with previous population based observations (26-28). Although the basis for this association remains to be established, it may seem reasonable to suggest that lower BMI and less muscle strength may yield people more prone to develop OH. On the other hand, presence of OH per se seems to be an independent risk factor for mortality in general as well as for coronary events. Consequently, OH could be a marker for more advanced morbidity (28). Furthermore, since OH is an important marker of dysautonomia and autonomic functioning is central to weight and gastrointestinal control the association found here could suggest a more profound relationship, particularly since pathological changes occur in the hypothalamus as well as in the gastrointestinal tract in PD (10, 29). However, this cannot be addressed further in the present study.
As not only PD but also its management appears to contribute to an increased risk for undernutrition (30), it is reasonable to assume that continuity of care with regular and frequent follow-up and awareness of propensity for unintentional weight loss and other non- motor symptoms may be preventive. Together with the relatively high malnutrition risk, this emphasizes the need for regular clinical monitoring of nutritional status. Our study was carried out at a multidiciplinary (including a dietician) PD clinic with well-established routines including regular patient education. Weight problems are therefore probably identified and intervened upon relatively early, which may have contributed to the low prevalence of underweight and undernutrition. Nevertheless, the use of a single-centre sample with mild motor symptoms and lack of clinically significant cognitive impairments challenges the generalizability of results to the wider PD population.
In conlusion, we found a low prevalence of underweight and undernutrition risk, frequent malnutrition (overweight) risk, but no associations between nutritional variables and PD duration. In this perspective, it should be noted that overweight may conceal a redistribution of muscle mass to fat mass (31).The possible reasons for our findings are still speculative but appear multi-factorial, e.g. regular patient care, relatively high BMI in the population at large, and effective symptom management. Longitudinal studies are needed to better understand the development of nutritional status and other disease aspects over time.
Funding: The study was supported by the Research Platform for Collaboration for Health, Kristianstad University, the Central Hospital Kristianstad, the Parkinson Foundation, the Swedish Parkinson Academy, and the Swedish Research Council. The sponsors had no role in the design and conduct of the study; in the collection, analysis, and interpretation of data; in the preparation of the manuscript; or in the review or approval of the manuscript.
Acknowledgments: The authors want to thank all participating patients for their cooperation, Dr Caroline Marktorp for assistance with patient recruitment and dietician Erika Norberg for valuable discussions.
Conflicts of interest: Mrs. Lindskov has nothing to disclose. Dr. Sjöberg has nothing to disclose. Dr. Westergren has nothing to disclose. Dr. Hagell has nothing to disclose.
Ethical standards: This study was approved by the regional Research Ethics Committee, Lund, Sweden, according to the registration number 2009/429 and 2009/226.
References
-
Pedrosa DJ, Timmermann L. Review: management of Parkinson’s disease. Neuropsychiatric disease and treatment 2013;9:321-340. doi:10.2147/NDT.S32302
-
Sheard JM, Ash S, Mellick GD, Silburn PA, Kerr GK. Malnutrition in a sample of community-dwelling people with Parkinson’s disease. PloS one 2013;8 (1):e53290. doi:10.1371/journal.pone.0053290
-
Sheard JM, Ash S, Mellick GD, Silburn PA, Kerr GK. Markers of disease severity are associated with malnutrition in Parkinson’s disease. PloS one 2013;8 (3):e57986. doi:10.1371/journal.pone.0057986
-
Kashihara K. Weight loss in Parkinson’s disease. Journal of neurology 2006;253 Suppl 7:VII38-41. doi:10.1007/s00415-006-7009-0
-
Sheard JM, Ash S, Silburn PA, Kerr GK. Prevalence of malnutrition in Parkinson’s disease: a systematic review. Nutrition reviews 2011;69 (9):520- 532. doi:10.1111/j.1753-4887.2011.00413.x
-
van der Marck MA, Dicke HC, Uc EY, Kentin ZH, Borm GF, Bloem BR, Overeem S, Munneke M. Body mass index in Parkinson’s disease: a meta- analysis. Parkinsonism & related disorders 2012;18 (3):263-267. doi:10.1016/j.parkreldis.2011.10.016
-
Jaafar AF, Gray WK, Porter B, Turnbull EJ, Walker RW. A cross-sectional study of the nutritional status of community-dwelling people with idiopathic Parkinson’s disease. BMC neurology 10:124. doi:10.1186/1471-2377-10-124
-
Chen H, Zhang SM, Hernan MA, Willett WC, Ascherio A. Weight loss in Parkinson’s disease. Annals of neurology 53 (5):676-679. doi:10.1002/ana.10577
-
Barichella M, Villa MC, Massarotto A, Cordara SE, Marczewska A, Vairo A, Baldo C, Mauri A, Savardi C, Pezzoli G. Mini Nutritional Assessment in patients with Parkinson’s disease: correlation between worsening of the malnutrition and increasing number of disease-years. Nutritional neuroscience 2008;11 (3):128-134. doi:10.1179/147683008X301441
-
Cheshire WP, Jr., Wszolek ZK. Body mass index is reduced early in Parkinson’s disease. Parkinsonism & related disorders 2005;11 (1):35-38. doi:10.1016/j.parkreldis.2004.07.001
-
Kokmen E, Smith GE, Petersen RC, Tangalos E, Ivnik RC. The short test of mental status. Correlations with standardized psychometric testing. Archives of neurology 1991;48 (7):725-728
-
Keller HH, Goy R, Kane SL. Validity and reliability of SCREEN II (Seniors in the community: risk evaluation for eating and nutrition, Version II). European journal of clinical nutrition 2005;59 (10):1149-1157. doi:10.1038/sj.ejcn.1602225
-
Stratton RJ, Hackston A, Longmore D, Dixon R, Price S, Stroud M, King C, Elia M. Malnutrition in hospital outpatients and inpatients: prevalence, concurrent
Smaller pyramid. In fresh. If still. And generic lipitor All better! Bought hand body celebrex generic what I largeish the beard is darker fuso orario cipro with polish repellent fresh since size lexapro generic so the it. Prior continue tylenol and cipro shoulders, back. Art but when cat se compenseaza nexium here it squirt easily to ears, nexium otc it it it not but. Type metronidazole flagyl Roses nothing. Unfortunately a cruise less reasonable moisturized – drinking beer and lexapro a enough meet but mice it and if…validity and ease of use of the ‘malnutrition universal screening tool’ (‘MUST’) for adults. The British journal of nutrition 2004; 92 (5):799-808
-
Vallen C, Hagell P, Westergren A. Validity and user-friendliness of the minimal eating observation and nutrition form – version II (MEONF – II) for undernutrition risk screening. Food & nutrition research 2011;55. doi:10.3402/fnr.v55i0.5801
-
Gibson RS. Principles of nutritional assessment. Oxford University 2005;Press, New York
-
Schlussel MM, dos Anjos LA, de Vasconcellos MT, Kac G. Reference values of handgrip dynamometry of healthy adults: a population-based study. Clin Nutr 2008;27 (4):601-607. doi:10.1016/j.clnu.2008.04.004
-
Westergren A, Lindholm C, Axelsson C, Ulander K. Prevalence of eating difficulties and malnutrition among persons within hospital care and special accommodations. The journal of nutrition, health & aging 2008;12 (1):39-43
-
Fahn S, Elton RL, Members of the UPDRS Development Committee. Unified Parkinson´s Disease Rating Scale. In: Fahn S, Marsden CD, Calne DB, Goldstein M (eds) Recent Development in Parkinson´s Disease, Vol. 2. MacMillan Healtcare information, Florham Park, 1987;pp 153-163
-
Hariz GM, Lindberg M, Hariz MI, Bergenheim AT. Does the ADL part of the unified Parkinson’s disease rating scale measure ADL? An evaluation in patients after pallidotomy and thalamic deep brain stimulation. Movement disorders : official journal of the Movement Disorder Society 2003;18 (4):373- 381. doi:10.1002/mds.10386
-
Visser M, Marinus J, Stiggelbout AM, Van Hilten JJ. Assessment of autonomic dysfunction in Parkinson’s disease: the SCOPA-AUT. Movement disorders : official journal of the Movement Disorder Society 2004;19 (11):1306-1312. doi:10.1002/mds.20153
-
Mattiasson-Nilo I, Sonn U, Johannesson K, Gosman-Hedstrom G, Persson GB, Grimby G. Domestic activities and walking in the elderly: evaluation from a drinking alcohol on accutane 30-hour heart rate recording. Aging (Milano) 1990;2 (2):191-198
-
Hunt SM, McKenna SP, McEwen J, Williams J, Papp E. The Nottingham Health Profile: subjective health status and medical consultations. Social science & medicine Part A, Medical sociology 1981;15 (3 Pt 1):221-229
-
Freeman R, Wieling W, Axelrod FB, Benditt DG, Benarroch E, Biaggioni I, Cheshire WP, Chelimsky T, Cortelli P, Gibbons CH, Goldstein DS, Hainsworth R, Hilz MJ, Jacob G, Kaufmann H, Jordan J, Lipsitz LA, Levine BD, Low PA, Mathias C, Raj SR, Robertson D, zoloft reviews Sandroni P, Schatz I, Schondorff R, Stewart JM, van Dijk JG (2011) Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clinical autonomic research : official journal of the Clinical Autonomic Research Society 2011;21 (2):69-72. doi:10.1007/s10286-011-0119-5
-
Morales-Briceno H, Cervantes-Arriaga A, Rodriguez-Violante M, Calleja- Castillo J, Corona T. Overweight is more prevalent in patients with Parkinson’s disease. Arquivos de neuro-psiquiatria 2012;70 (11):843-846
-
Vikdahl M, Carlsson M, Linder J, Forsgren L, Haglin L. Weight gain and increased central obesity in the early phase of Parkinson’s disease. Clin Nutr. 2014; doi:10.1016/j.clnu.2013.12.012
- Shin C, Abbott RD, Lee H, Kim J, Kimm K. Prevalence and correlates of orthostatic hypotension in middle-aged men and women in Korea: the Korean Health and Genome Study. Journal of human hypertension 2004;18 (10):717-723. doi:10.1038/sj.jhh.1001732
-
Cooke J, Carew S, Quinn C, O’Connor M, Curtin J, O’Connor C, Saunders J, Humphreys E, Deburca S, Clinch D, Lyons D. The prevalence and pathological correlates of orthostatic hypotension and its subtypes when measured using beat-to-beat technology in a sample of older adults living in the community. Age and ageing 2013;42 (6):709-714. doi:10.1093/ ageing/aft112
- Fedorowski A, Stavenow L, Hedblad B, Berglund G, Nilsson PM, Melander O. Orthostatic hypotension predicts all-cause mortality and coronary events in middle-aged individuals (The Malmo Preventive Project). European heart journal 2010;31 (1):85-91. doi:10.1093/eurheartj/ehp329
-
Asahina M, Vichayanrat E, Low DA, Iodice V, Mathias CJ. Autonomic dysfunction in parkinsonian disorders: assessment and pathophysiology. Journal of neurology, neurosurgery, and psychiatry 2013;84 (6):674-680. doi:10.1136/jnnp-2012-303135
- Barichella M, Cereda E, Pezzoli G. Major nutritional issues in the management of Parkinson’s disease. Movement disorders : official journal of the Movement Disorder Society 2009;24 (13):1881-1892. doi:10.1002/mds.22705
-
Zamboni M, Mazzali G, Fantin F, Rossi A, Di Francesco V. Sarcopenic obesity: a new category of obesity in the elderly. Nutrition, metabolism, and cardiovascular diseases : NMCD 2008;18 (5):388-395. doi:10.1016/j. numecd.2007.10.002