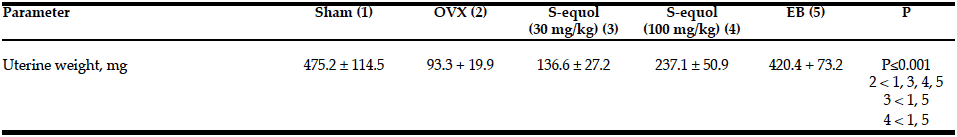J.A. Yu-Yahiro1, C.B. Ruff2, B.G. Parks1, V.S. Sinkov1, I. Merchenthaler3
1. Department of Orthopaedic Surgery, MedStar Union Memorial Hospital, Baltimore, MD; 2. Functional Anatomy and Evolution, Johns Hopkins School of Medicine, Baltimore, MD; 3. Epidemiology & Public Health and Anatomy/Neurobiology, University of Maryland School of Medicine, Baltimore, MD.
Corresponding Author: Janet A. Yu-Yahiro, Ph.D., MedStar Union Memorial Orthopaedics, The Johnston Professional Building, #400, 3333 North Calvert Street, Baltimore, MD 21218, 001-410-554-6668; fax 001-410-261-8105; e-mail: lyn.camire@medstar.net
Abstract
Background: To investigate the effect of S-equol, a selective estrogen receptor ß agonist produced in certain individuals by biotransformation of the soy isoflavone daidzein, on bone structure, bone strength, and metabolism in overiectomized rats. Design: Controlled animal study. Participants: Total of 75 female rats. Intervention: Animals were divided into 5 groups: ovariectomized (OVX), OVX+17-beta estradiol benzoate (EB), OVX+S-equol (30 mg/kg), OVX+S-equol (100 mg/kg), and SHAM. Animals received drug or vehicle for 60 days. At sacrifice, right femora and vertebrae (L3 and L4) were excised. Measurements: Bone density and structural parameters were measured by pQCT. Mechanical testing and quantitative histomorphometry were done. Blood markers of bone metabolism and uterine weights were measured. Results: Higher dose S-equol preserved mechanical strength of bone. Vertebral compressive strength, femoral bending strength, and femoral cortical thickness were not different between the S- equol (100 mg/kg) , SHAM, and EB groups and all were significantly higher than OVX and S-equol (30 mg/kg) groups. No differences were found in osteoclast numbers or vertebral bone mineral composition, and serum markers of bone metabolism did not follow the pattern of strength measures differences. Uterine weight in the higher dose S-equol group was significantly lower than in SHAM and EB groups. Conclusions: Treatment of OVX rats with S-equol (100 mg/kg) resulted in preservation of vertebral and femoral bone strength and volume not different from that in SHAM or EB rats. Higher dose S-equol caused less uterine stimulation than did endogenous or synthetic estrogen. These results suggest that S-equol warrants further study as a possible alternative to estrogen replacement for treatment of osteoporosis.
Key words: Mechanical loading, bone, bone turnover markers, bone histomorphometry.
Introduction
The consequences of osteoporosis and related osteoporotic fractures are devastating, both medically and economically. Today in the U.S. alone there are more than 2 million osteoporotic fractures per year that cost over $17 billion dollars annually to treat. It is estimated that by the year 2025, the number of fractures will rise by 50% and the cost of treating these fractures will exceed $25 billion per year (8). Between 16% and 32% of those who suffer a hip fracture in the U.S. die within the first year after fracture (4, 11). Many of those who do survive require assistive devices for walking and can perform fewer independent activities of daily living compared to before fracture (27).
The decrease in estrogen production during menopause is associated with long-term loss of bone mineral density and increased fracture rate. Though effective in preventing loss of bone mineral density after menopause, some of the existing anti-resorptive agents are associated with undesirable side effects including increased risk of ovarian and uterine cancer (24). Parathyroid hormone, an anabolic agent, causes increases in bone modeling and remodeling. However, it seems to exert its most beneficial effects in the early months of treatment and such effects appear to reverse upon cessation of treatment unless an additional antiresorptive agent such as bisphosponate is use given upon cessation (13). It would be desirable to identify other agents for treatment of osteoporosis resulting from a decrease in estrogen production.
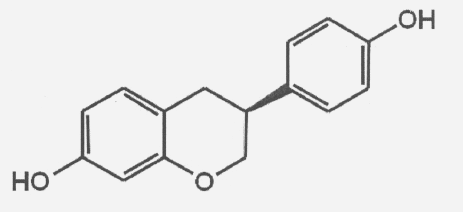
A number of reports suggest that soy, isoflavones contained in soy, and racemic equol improve bone quality and prevent loss of bone mineral density in animals and humans (12, 15, 19, 30, 35, 40), but studies are inconclusive about its mechanism of action. S-equol is an isoflavone produced by the bacterial biotransformation of daidzein, an isoflavone in soy, in some individuals (32). The S-enantiomer of equol, S-equol is a biologically active ceftin is used to treat compound that is an estrogen receptor ligand with 30-fold lower affinity for ER α than for ER ß (Figure 1) (23). In controlled clinical studies in postmenopausal women, daidzein consumption has been associated with improved bone mineral density (BMD) in those subjects who are equol producers (3, 17, 32, 40). Oral bioavailability of S-equol is high (82%) (33).
A recent double-blinded, randomized, controlled trial found that S-equol treatment of postmenopausal women who were not S-equol producers resulted in markedly inhibited bone resorption and no decrease in bone mineral density (36). No study has yet evaluated the clinical effects of pure S-equol on bone mechanical strength or on bone structure and formation. Our purpose was to compare various indicators of bone quality and bone strength in ovariectomized (OVX) rats receiving S- equol or estrogen treatment and in sham operated rats. We hypothesized that S-equol would maintain mechanical strength of the bones in a manner comparable to endogenous or synthetic estrogen and that measures of bone structure, formation, and resorption as measured by bone markers and quantitative histomorphometry in S- equol-treated rats would be similar to those found in sham operated or estrogen-treated OVX to diabetic patients. mon, 01/20/2014 – 6:36pm. should you would want to receive more rats.
Materials and methods
Materials
S-equol (purity, 98.8%) was obtained from Girindus Solvay America, Inc. (Cincinnati, OH, USA).
Animal Model
We obtained institutional animal care and use committee approval for the study from the University of Maryland Animal Care and Use Committee. Seventy-five female rats 60 days old were divided into 5 groups of 15 animals each: OVX, OVX with estrogen replacement (17- beta estradiol benzoate) 0.4 mg/kg (EB) (7), OVX with S- equol (30 mg/kg) or (100 mg/kg), and sham OVX (SHAM). S-equol was dissolved in DMSO to the particular dose with sesame oil such that the final concentration of DMSO was 5%. The OVX and SHAM groups were given DMSO/sesame oil. The amount of vehicle given to each rat was 0.5 ml. Treatment began on the day of surgery. Animals were treated for 60 days receiving daily oral doses of the drug or vehicle. All dosages were adjusted at 3-week intervals based on group mean body weight. All rats were fed a phytoestrogen-free diet (Teklad TD 2016, Harlan Laboratories, Indianapolis, IN) ad libitum. When the rats were 120 days old, they were euthanized by CO2 overdose based on National Institutes of Health guidelines. The right femur, lumbar vertebra (L3 and L4), and uterus of each specimen were collected.
Structural Parameters
Right femora and lumbar vertebrae were cleaned of soft tissue and frozen at -20° C. Structural parameters of these bones including geometry, mass, and density were measured by pQCT scans (Stratec Research, SA, Stratec Medizintechnik, Pforzheim, Germany) using a cross- sectional resolution of 0.1 mm and a slice thickness of 1 mm. pQCT scans of L3 were taken across two contiguous slices through the center of the body, and properties were averaged across the two slices. A threshold of 280 mg/cm³ was used to distinguish cortical and trabecular bone from air and marrow. Bone mineral content (BMC) and bone mineral density (BMD) of the two sections were determined and averaged. Femora were scanned at 45% of total bone length from the distal end. Anteroposterior (AP) section modulus (an index of bending strength) in the femur was determined using Stratec software.
Mechanical Testing
After pQCT scanning, L3 vertebrae were milled into approximately 4-mm axial sections with parallel cranial and caudal ends and with posterior elements and processes removed, using a Buehler isomet and fine bone saw (MicroAire), respectively. Biomechanical testing was carried out using an MTS servohydraulic load frame (Mini-bionix, MTS Systems, Inc.) following previously described protocols (14, 22). Vertebral specimens were tested in compression between parallel platens until failure at a loading rate of 2 mm/min. Femora were tested in 3-point bending, with the central loading point located at the same 45% section location as that used for pQCT measures, and specimens were loaded to failure at 2 mm/min. The orientation of the femur was anterior surface up to be contacted first by the loading point from above.
Because mechanical parameters are most relevant when expressed relative to body size (31), all mechanical and structural parameter data with the exception of densities were divided by body weight (grams) before statistical analysis. Measurements of vertebral BMC and peak compressive load were divided by body weight (grams), and femoral AP section modulus and peak bending load were avodart pills divided by body weight multiplied by bone length.
Bone Markers
At sacrifice, serum samples were collected, processed, and stored at -80C. Concentrations of osteocalcin and C- terminal telopeptide were determined in all groups. Both markers were measured in serum by enzyme-linked immunosorbent (ELISA) assay using kits manufactured by Nordic Bioscience Diagnostics. All samples were analyzed in duplicate, and controls were run throughout all assays.
Bone Histomorphometry
Undecalcified sections of vertebrae L4 (trabecular bone) and midshaft femur (cortical bone, 45% of total bone length from the distal end of the femur) were dehydrated by placing them sequentially in 5 containers of increasing concentrations of ethanol up to 100% ethanol for 24 h. They were then embedded in methylmethacrylate, sectioned, slide mounted, and stained using Mason-Goldner and VonKossa stains (18). Quantitative histomorphometric measurements of bone structure and resorption were determined using the Bioquant Image Analysis System (Bioquant Image Analysis System Corp., Nashville, TN) according to the manufacturer’s recommendations. All bone present in one section was clomid analyzed. Osteoclast numbers were derived from vertebral bone.
Statistical Analysis
Statistical analysis of data was done using a one-way ANOVA with pairwise post hoc Scheffe comparisons if ANOVA test yielded significant results. A p value of p ≤ 0.05 was accepted as significant. Bone parameter data were normalized based on body weight to avoid possible bias associated with the effect of increased weight on bone (1, 6, 34, 38).
Results
Body Weight
After 60 days of treatment, the OVX group had a significantly higher mean body weight (mean ± SD) of 300 ± 8 g versus 245 ± 11 g in the SHAM group (p=0.001). The EB, S-equol (30 mg/kg), and S-equol (100 mg/kg) groups had significantly lower mean body weights of 244 ± 14, 249 ± 28 and 255 ± 14 g, respectively, compared to the OVX group (p=0.01). The increase in weight associated with ovariectomy supported normalizing of the data based on weight as described above.
Bone Mineral Composition
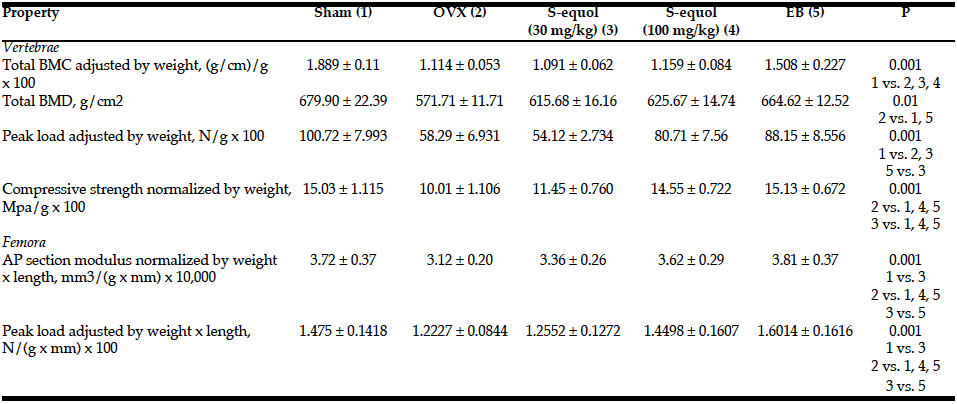
Adjusted BMC was significantly lower in the vertebrae of the OVX and both S-equol groups compared with the SHAM group (Table 1). Adjusted BMC in the EB group was not significantly different from that in the OVX or SHAM groups. Vertebral BMD was significantly lower in OVX compared to SHAM and EB groups; no other group differences reached significance.
Data are shown as mean ± SD. AP, anteroposterior; BMC, bone mineral composition; BMD, bone mineral density. P values: 1-way ANOVA at p value shown, followed by post-hoc Scheffe comparisons significant at p <.05.
Bone Mechanical Properties
Vertebral peak load adjusted for body weight was significantly lower in the OVX and lower S-equol dose (30 mg/kg) groups than in SHAM or EB groups (the latter with S-equol 30 mg/kg only) (Table 1). The higher dose S-equol (100 mg/kg), EB, and SHAM groups were not different in vertebral peak load. In the femur, AP section modulus and peak bending load adjusted for body weight and femoral length were significantly lower in OVX and the lower S-equol dose (30 mg/kg) groups than http://accutanegeneric-reviews.com/ in SHAM or EB groups. The higher S-equol dose (100 mg/kg) group had significantly greater femoral strength properties than OVX or lower S-equol dose groups and was not significantly different from the SHAM or EB groups (Table 1).
Bone Metabolism
The concentrations of osteocalcin and C-telopeptide in the SHAM group fell within the range described as normal by the manufacturer. Interassay variability for both assays was less than 2%. The SHAM group had significantly lower levels of C-telopeptide than all other groups, indicating less bone turnover than in the other groups (Table 2). Osteocalcin levels were significantly lower in the EB group than in all other groups. The S- equol higher dose (100 mg/kg) group had a 4.5-fold higher level of osteocalcin than the EB group.
Data are shown as mean ± SD. P values: 1-way ANOVA at p value shown, followed by post-hoc Scheffe comparisons significant at p <.05.
Histomorphometry
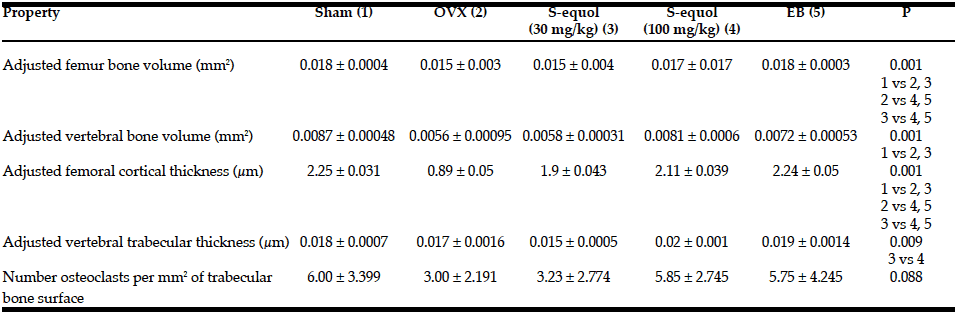
The higher dose S-equol (100 mg/kg) group showed weight-adjusted (divided by weight in grams) femoral and vertebral bone volume and femoral cortical thickness not different from that of the SHAM operated rats or those receiving estrogen, while the OVX and lower dose S-equol groups were lower in these parameters (Table 3). Vertebral sections showed no differences in trabecular thickness or number of osteoclasts per square millimeter of trabecular bone surface between SHAM, OVX, EB, and S-equol groups.
P values: 1-way ANOVA at P value shown, followed by zoloft online post hoc Scheffe comparisons significant at p <.05.
Uterotrophic Activity
Uterine weight in all groups was significantly greater than in the OVX group at the p=0.001 level (Table 4). Uterine weight in both S-equol groups was significantly lower than in the SHAM and EB groups (p=0.001).
Data are shown as mean ± SD. P values: 1-way ANOVA at p value shown, followed by post-hoc Scheffe comparisons.
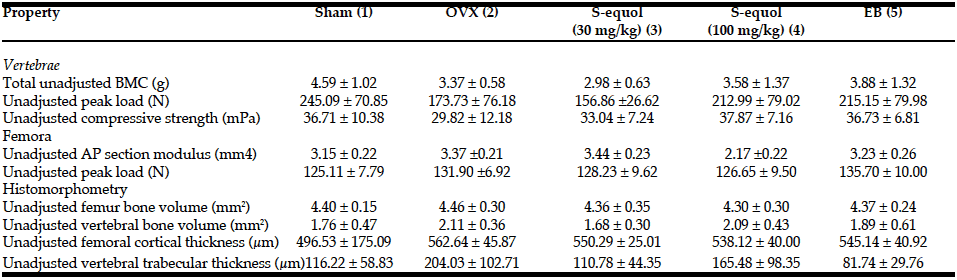
Unadjusted Data
Unadjusted data are shown in Table 5.
Data are shown as mean ± SD. BMC, bone mineral composition; AP, anteroposterior.
Discussion
Higher dose S-equol treatment prevented ovariectomy- induced losses of both femoral and vertebral strength. The OVX group had a femoral bending strength about 20% lower than in the SHAM group, but femoral bending strength in OVX rats treated with 100 mg of S-equol per kg was not different from that in the endogenous (SHAM) or synthetic (EB) estrogen groups. This effect on bone strength was seen even more dramatically in the vertebrae, where vertebral compressive peak load in OVX rats at sacrifice was only a little over half that of the intact (SHAM) control whereas the higher-dose S-equol group and the SHAM group were nonsignificantly different. Uterine weight in the current study was higher in rats treated with 100 mg of S-equol per kg than in the OVX group, but uterine weight in both S-equol groups was significantly lower than in the SHAM and EB groups. S- equol did not affect bone mineral density or osteoclastic or osteoblastic activity as measured by levels of serum osteocalcin and C-telopeptide or by the histomorphometric determination of osteoclast number. Strength measures done in this study cannot be replicated in women, but some investigators suggest that newly available noninvasive measures of bone geometry may be good predictors of bone strength (5).
Uterine stimulation with both doses of S-equol was significantly greater
than in the OVX group, but was significantly lower than in the endogenous or synthetic estrogen groups. Although uterine stimulation did occur, significantly less uterine stimulation with S-equol compared with estrogen may be an advantage of S-equol treatment. S-equol has a 30-fold lower affinity for ER α than for ER ß (23). S-equol may have a more favorable safety profile than estrogens that bind equally to ER α and ER ß, such as estradiol (23). The binding affinity of S- equol for ERß is 1000 times lower than that of estradiol (23). Estrogen replacement therapy for postmenopausal osteoporosis is limited because of the unwanted stimulation of estrogen receptors at extraskeletal sites such as the breast and possible increased risk of breast cancer (29).
Selective estrogen receptor modulators (SERMs) have been developed to selectively stimulate only estrogen receptors in a tissue-specific manner with beneficial effects, and several are commercially available. They are thought to act by causing tissue-specific conformational changes in the estrogen receptor that change the affinity of the estrogen receptor to its ligand (20). S-equol, a soy- based phytoestrogen, may work in a similar fashion to SERMs by binding to ERs. However, it is a conundrum that in this study, S-equol, primarily an ER ß agonist, had an effect on bone, which is considered primarily an ER α target (26). This finding suggests that S-equol utilizes other yet unknown, perhaps ER-independent mechanisms to protect the bone in hypoestrogenic conditions. The significantly lower effect of S-equol versus estradiol on the uterus, another primarily ER α target (26), further suggests that ER mechanisms do not fully explain the action of S-equol. Although both ER α and ER ß receptors are present in uterine and mammary tissue, ER ß agonists alone appear to have little effect on these tissues (26). In contrast, racemic equol, a commercially available form of equol that contains 50% S- equol and 50% R-equol, has shown beneficial effects on bone (12, 16, 30, 35) but has a 25% higher affinity for ER α compared with ERß and has been associated with estrogen stimulation at extraskeletal sites (23).
Weight-adjusted data were used for calculations involving effects of treatment on structural changes of the bone. Numerous clinical studies have used normalization based on body mass index or body weight to avoid bias where larger body size would have an independent effect on bones (1, 6, 34, 38). Preclinical studies have also normalized data based on body weight where changes to bone are being studied (16, 35). The confounding effect of larger size can be seen in our unadjusted raw data for peak load, where peak femur load in the estrogen group was significantly higher than in the SHAM operated group. When data were adjusted for weight, peak load in the estrogen group was not significantly different from that in the SHAM operated group. We provided the raw data for reference, but we analyzed the adjusted data.
Our findings of higher vertebral compressive and femoral bending strengths with S-equol treatment are consistent with previous reports using animal models. Sehmisch et al. (30) reported higher vertebral compressive strength, Tezval et al. (35) found higher trochanteric breaking strength, and Mathey et al. (19) reported higher femoral load in rats fed diets containing racemic equol for 5 weeks compared to OVX rats receiving a soy-free diet over the same time period. Effects of treatment on other parameters, including BMC and BMD, are less consistent. In the current study, neither dose of S-equol had a beneficial effect on vertebral BMC (adjusted for body size) or on BMD measured by pQCT. Rachon (28) treated rats for 6 weeks with racemic equol and also found no improvements in BMD of either the tibia or lumbar spine by CT. In contrast, Kim et al. (15) and Fujioka et al. (12) in the mouse model, and Mathey et al.[19] and Tsuang et al. (37) in the rat model found significant increases in BMD of either the femur, lumbar spine, or whole body compared to OVX controls after treatment with racemic equol or soy isoflavones.
Weight-adjusted femoral and vertebral bone volume and femoral cortical thickness in those rats receiving higher dose S-equol in the current study were no different than in the SHAM and estrogen-treated groups, and these parameters in all three groups were significantly higher than in the OVX controls. Tezval et al. (35) also found that racemic equol-treated rats had significantly higher trabecular area and connectivity compared to an OVX group but there was no difference in number of osteoclasts among groups. In contrast, Fujoika et al. (12) found significantly fewer osteoclasts in the rats treated with racemic equol compared to OVX rats.
Bone mineral density and all of the factors comprising bone quality such as bone geometry, bone microarchitecture, the quality of the collagen, and the rate of bone turnover are important determinants of how well a skeleton can resist fracturing (2, 9, 10). The most important factor or combination of factors for bone preservation has not yet been determined, and studies have shown that existing treatments for osteoporosis target some but not all of these factors (25). The current data show that S-equol may be promising in its ability to impact at least some of these functionally critical parameters.
Our methodology allowed identification of key data, but µCT and TRAP staining would have provided additional detail about bone strength and osteoclast surface and numbers. Also, we were restricted to using 60-day old rats in this study. Other studies have used older rats for osteoporotic rat models, but some investigators have used rats of approximately the same age as ours for similar models (21, 39).
Conclusions
In conclusion, treatment of OVX rats with S-equol (100 mg/kg) resulted in vertebral and femoral bone strength and bone volume not different from that in sham- operated or estrogen-treated rats without change in bone density. S-equol in both doses caused less uterine stimulation than either endogenous or synthetic estrogen.
These results suggest that S-equol warrants further study as a possible alternative to estrogen replacement for treatment of osteoporosis.
Acknowledgments: The study was funded by a research contract from Ausio Pharmaceuticals. The funding institution had no other role in the design, conduct, or reporting of the study. The authors had full access to all data, performed all analyses, and interpreted all data independently of the funding organization. We gratefully acknowledge Richard Jackson and Richard Schwen of Ausio Pharmaceuticals for donation of pure S-equol and for input on dosages, route of administration, and length of treatment. We are grateful to Sione Fanua, M.S., for expert care of experimental animals and surgery assistance, to Jeff Yahiro for bone histomorphometry, and to Lyn Camire, MA, ELS, of MedStar Union Memorial Orthopaedics for editorial assistance.
Disclosures: The study was funded by Ausio Pharmaceuticals. The funding institution had no other role in the design, conduct, or reporting of the study.
Conflicts of interest: The authors report no conflict of interest associated with this study.
Ethical standards: This manuscript is in accord with the journal’s publication ethics statement.
References
1. Alele JD, Kamen DL, Hunt KJ, Ramsey-Goldman R. Bone geometry profiles in women with and without SLE. J Bone Miner Res 2011;26:2719-2726.
2. Ammann P, Rizzoli R. Bone strength and its determinants. Osteoporos Int 2003;14 Suppl 3:S13-S18.
3. Atkinson C, Frankenfeld CL, Lampe JW. Gut bacterial metabolism of the soy isoflavone daidzein: exploring the relevance to human health. Exp Biol Med (Maywood ) 2005;230:155-170.
4. Bass E, French DD, Bradham DD, Rubenstein LZ. Risk-adjusted mortality rates of elderly veterans with hip fractures. Ann Epidemiol 2007;17:514-519.
5. Beck TJ, Kohlmeier LA, Petit MA, Wu G, LeBoff MS, Cauley JA, Nicholas S, Chen Z. Confounders in association between exercise and femur bone in postmenopausal women. Med Sci Sports Exerc 2011;43:80-89.
6. Biewener AA, Bertram JE. Structural response of growing bone to exercise and disuse. J Appl Physiol 1994;76:946-955.
7. Bottner M, Christoffel J, Wuttke W. Effects of long-term treatment with 8- prenylnaringenin and oral estradiol on the GH-IGF-1 axis and lipid metabolism in rats. J Endocrinol 2008;198:395-401.
8. Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res 2007;22:465-475.
9. Chavassieux P, Seeman E, Delmas PD. Insights into material and structural basis of bone fragility from diseases associated with fractures: how determinants of the biomechanical properties of bone are compromised by disease. Endocr Rev 2007;28:151-164.
10. Clarke B. Normal bone anatomy and physiology. Clin J Am Soc Nephrol 3 Suppl 2008;3:S131-S139.
11. Deakin DE, Boulton C, Moran CG. Mortality and causes of death among patients with isolated limb and pelvic fractures. Injury 2007;38:312-317.
12. Fujioka M, Uehara M, Wu J, Adlercreutz H, Suzuki K, Kanazawa K, Takeda K, Yamada K, Ishimi Y. Equol, a metabolite of daidzein, inhibits bone loss in ovariectomized mice. J Nutr 2004;134:2627.
13. Gallacher SJ, Dixon T. Impact of treatments for postmenopausal osteoporosis (bisphosphonates, parathyroid hormone, strontium ranelate, and denosumab) on bone quality: a systematic review. Calcif Tissue Int 2010;87:469-484.
14. Gerharz EW, Mosekilde L, Thomsen JS, Gasser JA, Moniz C, Barth PJ, Ransley PG, Woodhouse CR (2003) The effect of enterocystoplasty on bone strength assessed at four different skeletal sites in a rat model. Bone 2003;33:549-556.
15. Kim DW, Yoo KY, Lee YB, Lee KH, Sohn HS, Lee SJ, Cho KH, Shin YK, Hwang IK, Won MH, Kim DW. Soy isoflavones mitigate long-term femoral and lumbar vertebral bone loss in middle-aged ovariectomized mice. J Med Food 2009;12:536-541.
16. Kolios L, Sehmisch S, Daub F, Rack T, Tezval M, Stuermer KM, Stuermer EK. Equol but not genistein improves early metaphyseal fracture healing in osteoporotic rats. Planta Med 2009;75:459-465.
17. Lydeking-Olsen E, Beck-Jensen JE, Setchell KD, Holm-Jensen T. Soymilk or progesterone for prevention of bone loss–a 2 year randomized, placebo- controlled trial. Eur J Nutr 2004;43:246-257.
18. Malluche HH, Faugere MC. Evaluation of mineralized bone histology. In: Malluche HH, Faugere MC (eds) Atlas of Mineralized Bone Histology, Karger, Basel, Switzerland, 2006;pp 37-48
19. Mathey J, Mardon J, Fokialakis N, Puel C, Kati-Coulibaly S, Mitakou S, netau- Pelissero C, Lamothe V, Davicco MJ, Lebecque P, Horcajada MN, Coxam V. Modulation of soy isoflavones bioavailability and subsequent effects on bone health in ovariectomized rats: the case for equol. Osteoporos Int 2007;18:671- 679.
20. McDonnell DP, Clemm DL, Hermann T, Goldman ME, Pike JW. Analysis of estrogen receptor function in vitro reveals three distinct classes of antiestrogens. Mol Endocrinol 1995;9:659-669.
21. Miao Q, Li JG, Miao S, Hu N, Zhang J, Zhang S, Xie YH, Wang JB, Wang SW. The bone-protective effect of genistein in the animal model of bilateral ovariectomy: roles of phytoestrogens and PTH/PTHR1 against post- menopausal osteoporosis. Int J Mol Sci 2012;13:56-70.
22. Mosekilde L, Thomsen JS, Orhii PB, McCarter RJ, Mejia W, Kalu DN. Additive effect of voluntary exercise and growth hormone treatment on bone strength assessed at four different skeletal sites in an aged rat model. Bone accutane reviews 1999;24:71-80.
23. Muthyala RS, Ju YH, Sheng S, Williams LD, Doerge DR, Katzenellenbogen BS, Helferich WG, Katzenellenbogen JA. Equol, a natural estrogenic metabolite from soy isoflavones: convenient preparation and resolution of R- and S-equols and their differing binding and biological activity through estrogen receptors alpha and beta. Bioorg Med Chem 2004;12:1559-1567.
24. Neves E Castro M. Association of ovarian and uterine cancers with postmenopausal hormonal treatments. Clin Obstet Gynecol 2008;51:607-617.
25. Nikander R, Sievanen H, Heinonen A, Daly RM, Uusi-Rasi K, Kannus P. Targeted exercise against osteoporosis: A systematic review and meta- analysis for optimising bone strength throughout life. BMC Med 2010;8:47.
26. Nilsson S, Gustafsson JA. Estrogen receptors: therapies targeted to receptor subtypes. Clin Pharmacol Ther 2011;89:44-55.
27. Office of Technology Assessment. Hip fracture outcomes of people age 50 and over–background paper, OTA-BP-H-120, 1994.
28. Rachon D, Seidlova-Wuttke D, Vortherms T, Wuttke W. Effects of dietary equol administration on ovariectomy induced bone loss in Sprague-Dawley rats. Maturitas 2007;58:308-315.
29. Rossouw JE, Anderson GL, Prentice RL, LaCroix AZ, Kooperberg C, Stefanick ML, Jackson RD, Beresford SA, Howard BV, Johnson KC, Kotchen JM, Ockene J. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 2002;288:321-333.
30. Sehmisch S, Erren M, Kolios L, Tezval M, Seidlova-Wuttke D, Wuttke W, Stuermer KM, Stuermer EK. Effects of isoflavones equol and genistein on bone quality in a rat osteopenia model. Phytother Res 24 Suppl 2010;2:S168- S174.
31. Selker F, Carter DR. Scaling of long bone fracture strength with animal mass. J Biomech 1989;22:1175-1183.
32. Setchell KD, Clerici C, Lephart ED, Cole SJ, Heenan C, Castellani D, Wolfe BE, Nechemias-Zimmer L, Brown NM, Lund TD, Handa RJ, Heubi JE. S- equol, a potent ligand for estrogen receptor beta, is the exclusive enantiomeric form of the soy isoflavone metabolite produced by human intestinal bacterial flora. Am J Clin Nutr 2005;81:1072-1079.
33. Setchell KD, Zhao X, Shoaf SE, Ragland K. The pharmacokinetics of S-equol administered as SE5-OH tablets to healthy postmenopausal women. J Nutr 2009;139:2037-2043.
34. Szulc P, Boutroy S, Vilayphiou N, Chaitou A, Delmas PD, Chapurlat R. Cross-sectional analysis of the association between fragility fractures and bone microarchitecture in older men: the STRAMBO study. J Bone Miner Res 2011;26:1358-1367.
35. Tezval M, Sehmisch S, Seidlova-Wuttke D, Rack T, Kolios L, Wuttke W, Stuermer KM, Stuermer EK. Changes in the histomorphometric and biomechanical properties of the proximal femur of ovariectomized rat after treatment with the phytoestrogens genistein and equol. Planta Med 2010;76:235-240.
36. Tousen Y, Ezaki J, Fujii Y, Ueno T, Nishimuta M, Ishimi Y. Natural S-equol decreases bone resorption in postmenopausal, non-equol-producing Japanese women: a pilot randomized, placebo-controlled trial. Menopause 18:563-574.
37. Tsuang YH, Chen LT, Chiang CJ, Wu LC, Chiang YF, Chen PY, Sun JS, Wang CC. Isoflavones prevent bone loss following ovariectomy in young adult rats. J Orthop Surg Res 2008;3:12.
38. Walker MD, Liu XS, Stein E, Zhou B, Bezati E, McMahon DJ, Udesky J, Liu G, Shane E, Guo XE, Bilezikian JP. Differences in bone microarchitecture between postmenopausal Chinese-American and white women. J Bone Miner Res 2011;26:1392-1398.
39. Wang J, Shang F, Mei Q, Wang J, Zhang R, Wang S. NO-donating genistein prodrug alleviates bone loss in ovariectomised rats. Swiss Med Wkly 2008;138:602-607.
40. Wu J, Oka J, Ezaki J, Ohtomo T, Ueno T, Uchiyama S, Toda T, Uehara M, Ishimi Y. Possible role of equol status in the effects of isoflavone on bone and fat mass in postmenopausal Japanese women: a double-blind, randomized, controlled trial. Menopause 2007;14:866-874.