B. Phillips, S. Ali, M. Chopra
IBBS, School of Pharmacy and Biomedical Science University of Portsmouth, Portsmouth PO1 2DT
Corresponding Author: Dr Mridula Chopra, School of Pharmacy and Biomedical Sciences, St Michael’s Building, White Swan Road, University of Portsmouth, Portsmouth PO1 2DT. United Kingdom, Tel: 02392842796; Fax:02392843565; Email: Mridula.Chopra@port.ac.uk
Abstract
Objectives: Benefits of physical activity on maintenance of blood glucose within an acceptable range are well documented. This study explores the possible beneficial effect
of animated conversation and mental activities on blood glucose levels following dietary carbohydrate loading. Design and Participants: Blood glucose changes were examined in 18 non-diabetic individuals following the consumption of two doughnuts providing ~48g of carbohydrate. The participants acted as their own controls and were studied twice. On the first occasion they maintained quiet/passive behaviour, on the second they conversed and participated in structured mental activities e.g. reading and solving puzzles. Measurements: Using the Freestyle Freedom Lite glucose metre, baseline, then post-fasting blood glucose concentrations were evaluated every 30 minutes over a 2 hour period after the consumption of doughnuts. Results: ANOVA repeated measures analysis of the results showed that time (P
Key words: Diabetes, glucose, quiet, animated conversation, mental activities.
Abbreviations: ISO: Organization for Standardization; IBBS: Institute of Biomedical and Biomolecular Research.
Introduction
Maintaining blood glucose homeostasis within a healthy range is important for health. Several factors can influence the glycaemic response of an individual following a carbohydrate loading. These include the glycaemic index of foods, co-ingestion of other macronutrients such as protein and fibre as well as engaging in physical activity (1-3). The effect of physical activity on glycaemic response is well known and a variety of activities can be just as valuable as performing one type of exercise (4). Although regular moderate aerobic exercise, alongside lifestyle interventions are beneficial for health, a lack of time means that many people do not fully comply with the guidelines and recommendations. There may also be times when exercise is not convenient or feasible due to a number of factors e.g. disability, illness, weather, work commitments or time constraints. Lack and inability to engage in physical activity can also be an issue for the elderly. In such situations alternative activities which are likely to increase glucose usage may be an option.
The effect of a relatively non-physical activity such as verbal communication on glycaemic response, following a carbohydrate load has not been examined before. Benefits of engaging in social activities and solitary reading are well documented for their physical and psychological benefits (5-7). The present study was set out to examine whether simple activities such as animated conversation combined with mental activities such as reading and/or solving puzzles could influence blood glucose changes following a carbohydrate load. The glycaemic response of participants was determined using a finger prick blood sample analysed by a glucose metre.
The accuracy and precision of glucose metres are known to vary and in some studies not all metres have been found to meet the minimum accuracy criteria of the WHO’s Organization for Standardization ISO 15197-2003 (8-10). The ISO standard for quality of capillary blood sample analysis with a glucose metre states that at glucose concentrations of
Methods
Ethical approval for this study was obtained from the Biosciences Research Ethics Committee of the University. The research was conducted in compliance with the declaration of Helsinki, as adopted at the 18th World Medical Association (WMA) General Assembly, Helsinki, Finland, 1964 and last amended at the 59th World Medical Association General Assembly, Seoul 2008.
Individuals with known diabetes, cardiovascular disease, gastrointestinal problems and bleeding disorders were excluded from participating. Individuals on any medication that was likely to influence glucose metabolism proper warm up before exercising and cool down afterward is essential to avoid injury and achieve peak performance i.e. Baclofen, Corticosteroids, L-dopa, and Tolazamide were also excluded from the study.
Metre comparison
Four hand held glucose metres were compared for their blood glucose measurements with the laboratory values of glucose. Metres that were used included:- Freestyle Freedom Lite (Abbott Diabetes Care, UK), Contour XT (Bayer Diabetes Care, UK), Accu-Chek Aviva (Roche Diagnostics Ltd, Accu-Chek UK) and One Touch Ultra 2 (LifeScan UK and Ireland). The reference laboratory value for glucose was obtained using Randox GLUC-PAP kit.
Thirty-two participants (13 male:19 female), age range 22-65 years were asked to provide 5 mL of venous blood as well as finger prick blood samples for glucose analysis. Participants were asked to wash their hands with warm water and their finger prick blood samples were obtained using One touch ultra soft lancets. Fingers were gently
squeezed to obtain the blood sample. The first drop of blood was wiped clean and the subsequent drops were used to determine the blood glucose values by four metres. Venous blood samples were obtained in fluoride oxalate tubes, plasma samples were obtained after centrifugation at 3000 rpm for 10 minutes and glucose concentrations were measured using Randox GLUC-PAP kit.
Doughnut Study
Twenty participants (8 male:12 female) gave their consent for participation and those with fasting blood glucose >6.0mmol/L (1 male, 1 female) were excluded from the study. Eighteen participants (7 male:11 female) age range 22-64 years (mean age ~ 40 ± 13), BMI 24.8 ± 3.3 kg/m2 (range 21-30) completed the study. Participants were advised to eat their evening meal at the same time of the day before each intervention and to maintain a similar exercise regime. After the evening meal, subjects were asked to fast overnight and to provide a fasting finger-prick blood sample the following morning. Prior to blood sampling, participants were asked to wash their hands with warm water and samples were obtained from the side of the fingers of the left hand. The first drop of blood was wiped off and subsequent drops were analysed using the Freestyle Freedom Lite metre. Duplicate measurements were performed at each time point.
Participants were asked to consume two doughnuts (sugar glazed, Morrisons, UK) providing ~48g of carbohydrate, then finger-prick blood samples were obtained at 30 minute intervals over a 2 hour period. Time intervals were determined using a timer after the last bite had been swallowed (note: participants were advised to maintain a similar chewing frequency for both interventions and it ranged from 12-16 chews/bite). The participants were then asked to either a) sit quietly for 2 hours or b) engage in non-physical activities such as animated conversation and/or problem solving or reading. Most volunteers however spent more time talking than on reading or problem solving. Overall, only about 25% of the time (20-30 minutes) was spent on reading and problem solving, the rest of the time was spent engaged in conversation. Each participant acted as their own control. Most participants completed both interventions within a week except for four participants, who had a gap of 2-3 weeks between the two interventions.
Data was compared using IBM SPSS for windows software (PASW18) with significance of difference set at P
Results
Metre accuracy and precision
Except for one touch ultra 2 metre which showed a precision of ~9% on six replicate measurements, all remaining three meters showed a precision of
i.e ≥5.6 mmol/l, only Accu Chek metre showed glucose concentrations significantly higher than the reference values (P P
Doughnut intervention
Blood glucose concentrations increased and peaked at 60 minutes in both interventions. Repeated measures ANOVA showed a significant effect of time (P=0.000) and activity (P=0.006) and a significant interaction between time and activity (P=0.04). Figure 1 shows that except for the baseline measurement, there is a clear difference in blood glucose levels when participants observed quiet/passive behaviour compared to when they were interactive. Figure 2 shows the change in blood glucose concentrations after correction for the baseline level. The change in blood glucose concentration was ~30-38% lower in the ‘active’ subjects, even at the first measurement at 30 minutes (Figure 2). Repeated measures ANOVA showed a significant effect of time at P value of 0.002 and activity at P=0.01, however interaction between time and activity was not significant when analysis was run on the differences.
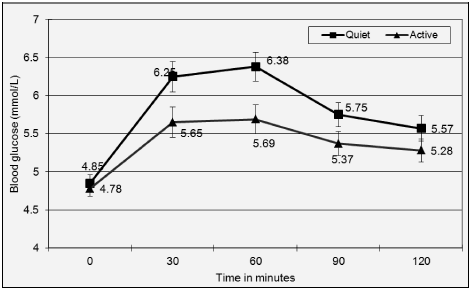
Figure 1: Comparison of the mean blood glucose concentrations (mmol/L) at 5 time points for each study period
a. Blood glucose values in mmol/l were measured in duplicate using Freestyle Freedom Lite meter. Values shown are mean and standard error obtained for quiet and active intervention at 5 time points.
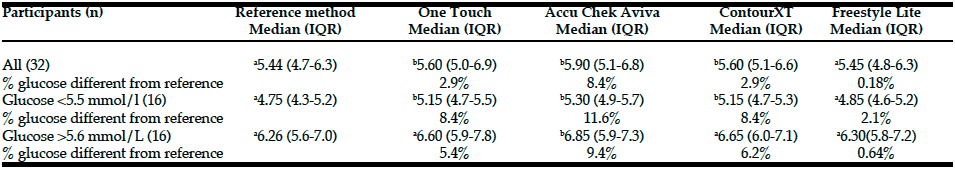
Table 1: Comparison of finger-prick blood glucose concentrations (mmol/L) from 4 different glucose metres with the laboratory reference method
Median and interquartile range of glucose values obtained with four metres (One touch ultra2, Accu Chek Aviva, ContourXT and Freestyle Freedom Lite) were compared with the venous blood glucose concentrations determined by the laboratory reference method (Randox GLUC-PAP). Percent deviation from the reference values was calculated for each metre. Significance of difference from the laboratory reference method was calculated using Wilcoxon paired sample test. Within each row, (a). that values are not significantly different from the reference values and (b). a significant difference at P
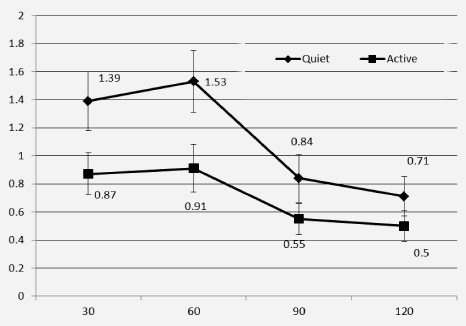
Figure 2: Change in blood glucose concentrations (mmol/L) for each study period after correction for baseline glucose concentrations
b. Baseline glucose concentrations (mmol/l) were subtracted from glucose concentrations all time points and change in blood glucose levels is shown for 30, 60, 90 and 120 minutes for the quiet and active intervention.
Discussion
In agreement with previously published reports (9) the Freestyle Freedom Lite glucose metre met the ISO standards most reliably, and although using a different principle of glucose analysis (glucose dehydrogenase) than the laboratory reference method (glucose oxidase) was found to be the most accurate in comparison (9,11).
However, all glucose metres gave slightly higher blood glucose concentrations than the laboratory reference method, especially at low concentrations. The Freestyle Freedom lite, ContourXT and Accu Check Aviva metres all use the glucose dehydrogenase method for blood glucose determination. The One Touch ultra2 glucose metre uses the glucose oxidase enzyme and gave results, which were less accurate. The methods differ in their specificity for glucose. Glucose oxidase with its cofactor flavin adenine dinucleotide (FAD) is very specific for glucose but interference can come from mannose. However it has been suggested that this interference only exists at very high mannose concentrations and is unlikely to be the case in real life (12). Medications containing non-glucose sugars (e.g. xylose and maltose) can interfere with the glucose dehydrogenase enzyme with its cofactor pyrroloquinoline quinine (13, 14). Therefore sugars other than glucose can cause potential errors in blood glucose measurements but were unlikely the case in the present investigation.
To our knowledge this is the first study that has shown that a non-physical activity such as animated conversation combined with reading and/or problem solving activity can lower the blood glucose concentration following a carbohydrate loading. Although there appear to be no studies on the effect of animated conversation on blood glucose levels, several studies have examined the effect of cognitive demand on blood glucose levels (15-18). One study found that the performance of cognitively demanding tasks was improved following a glucose drink, and the decline in blood glucose levels were dependent on the type of cognitive task, with liquid weed the reduction in blood glucose concentrations being greatest when the tasks were more demanding (18). Although the participants in the present study only spent 20-30 minutes on tasks that required focussed mental activity i.e. reading newspapers/ magazines or problem solving, they did spend the rest of the study in animated conversation which must also have required mental agility. We did find in our preliminary experiments (data not shown) that quiet conversation was not as effective at lowering the blood glucose concentrations as talking loudly, suggesting that just as the type of cognitive task influences the extent of blood glucose changes, the ‘type’ of talking will also influence blood glucose levels. Hence, not only physical exercise and mental activity as found by Scholey et al (18) but other activities such as animated conversation can also lower blood glucose concentrations. Perhaps such simple lifestyle interactions can be explored further for its benefits in patients with type 2 diabetes.
Various groups have researched the effect of physical activity on glycaemic control and quality of life in patients with type 2 diabetes. Many have confirmed improvements with regular exercise. A low intensity exercise such as arm swinging, 30 minutes a day, 3 days/week in patients with type 2 diabetes was shown to improve their glycaemic control (19). Two studies explored the effects of a home based physical exercise program and concluded that such a program has the potential for improvements in the quality of life, glycaemic control and weight loss in these patients (20, 21). The latter study also included a questionnaire about the barriers to physical exercise and highlighted that pain after physical activity, a lack of motivation and a low perception of capabilities for activity prevented individuals from adhering to their regime (21). Perseverance to an exercise program therefore can be a barrier that may prevent participation in regular exercise (22). This can be especially an issue for older adults (23). Our study has highlighted another factor that can be explored for its benefit in maintaining blood glucose levels within a healthy range. If interactive behaviour such as talking or engaging in mental activity can help patients control their blood glucose levels, then this may be a regime that patients can follow when they are unable to engage in regular exercise due to personal and environmental constraints.
There are many variables that can influence the outcome of an experiment such as the present one, for example, it has been suggested that the degree of mastication can influence the glycaemic response of an individual (24, 25). Participants in this study were therefore asked to keep a note of their chewing frequency during the first study period and were asked to maintain the same rate in the second to minimise its influence. Diet and lifestyle preceding the intervention may also affect the results. Dekker et al (26) found that the daily intake of caffeine over a two week period by caffeine naïve young males showed alterations to glucose homeostasis with increased serum insulin and blood glucose concentrations in response to the 5 mg caffeine/kg body weight/day. Exercise has also been shown to influence insulin sensitivity as studied by O’Donovan et al (27) who reported that 24 weeks of exercise can reduce insulin resistance and that both moderate or high intensity exercise can result in an improvement in insulin sensitivity of cells. A detailed record of the diet and exercise regime of participants at least 2-3 days preceding the intervention should have been taken as regular exercise of three times per week (27) or a one off exercise on the previous day of intervention (28) have both been shown to affect the insulin sensitivity of cells. We advised all our participants to eat their evening meal at the same time before each intervention and also maintain a similar exercise regime; and except for four, all participants completed the two interventions within 6 days. It is therefore unlikely to influence the results of our investigations. However the participants were not asked to provide a record of their diet and lifestyle preceding the experiment. This is something that should be considered for the future studies.
In conclusion, our investigation has highlighted a simple lifestyle change that can significantly influence blood glucose levels. We have shown that it is important to assist those unable to take part in physical activities to interact with other people and to encourage them to undertake mental activities and that such small change could assist in the control of blood glucose concentrations.
Acknowledgements: Authors would like to thank all volunteers without whose contribution this research could not have been completed. Authors would also like to thank Mrs Blerta Jakaj and Mrs Sonali Chopra for their assistance with the doughnut intervention study. This work was funded by IBBS, University of Portsmouth.
Authorship: All authors took part in the doughnut intervention experiment. Glucose comparison of pin prick samples with the laboratory results was undertaken by Syed and Beverly. All authors confirm there is no conflict of interest. Ethical approval for this study was obtained from the Biosciences Research Ethics Committee of the University. The research was conducted in compliance with the declaration of Helsinki, as adopted at the 18th World Medical Association (WMA) General Assembly, Helsinki, Finland, 1964 and last amended at the 59th World Medical Association General Assembly, Seoul 2008.
References
1. Collier G, McLean A, O’Dea K. Effect of co-ingestion of fat on the metabolic responses to slowly and rapidly absorbed carbohydrates. Diabetologia 1984;26, 50 –55.
2. Krishnan S, Rosenberg L, Singer M et al. Glycemic index, glycemic load, and cereal fiber intake and risk of Type 2 Diabetes in US black women. Arch Intern Med 2007;167(21), 2304-2309.
3. Mayer-Davis EJ, D’Agostino R Jr, Karter AJ et al. Intensity and amount of physical activity in relation to insulin sensitivity: the Insulin Resistance Atherosclerosis Study. JAMA 1998;279, 669-674.
4. Sanz C, Gautier JF, Hanaire H. Physical exercise for the prevention and treatment of type 2 diabetes. Diabetes Metab 2010;36 (5), 346-351.
5. Menec VH. The relation between everyday activities and successful aging: a 6-year longitudinal study. J Gerontol B Psychol Sci Soc Sci 2003;58(2):S74-S82.
6. Cohen S. (2004) Social relationships and Health. Am Psychol 59 (8):676-684.
7. Gautam R, Saito T and Kai I. Leisure and religious activity participation and mental health: gender analysis of older adults in Nepal. BMC Public Health 2007;7:299-320.
8. Kuo CY, Hsu CT, Ho CS et al. Accuracy and precision evaluation of seven self-monitoring blood glucose systems. Diabetes Technol Ther 2011;13(5), 596-600.
9. Tack C, Pohlmeler H, Behnke T et al. Accuracy evaluation of five blood glucose monitoring systems obtained from the Pharmacy: A European Multicenter Study with 453 Subjects. Diabetes Technol Ther 2012;14 (4), 330–337.
10. Hellman R. Glucose meter inaccuracy and the impact on the care of patients. Diabetes Metab Res Rev 2012;28 (3), 207-209.
11. Zeuger T, Schuler V, Stettler C et al. Assessment of three frequently used blood glucose monitoring devices in clinical routine. Swiss Med Wkly, 2012;42, w13631.
12. Hinzman R, Tran C-T. System accuracy evaluation for blood glucose measurements should be performed with ISO 15197:2003 and CLSI-approved guideline EP7-A2. Diabetes Technol Ther 2012;4(4), 379-380.
13. Montagnana M, Caputo M, Giavarina D et al. Overview on self-monitoring of blood glucose. Clin Chim Acta, 2009;402 (1-2): 7 – 13.
14. Vashist SK, Zheng D, Al-Rubeaan K, Luong J. Technology behind commercial devices for blood glucose monitoring in diabetes management: A review. Anal Chim Acta 2011;703 (2), 124 – 136.
15. Donohoe RT, Benton D. Glucose tolerance predicts performance on tests of memory and cognition. Physiol Behav 2000;71 (3-4): 395 – 401.
16. Martin PY, Benton D. The influence of a glucose drink on a demanding working memory task. Physiol Behav 1999;67 (1): 69 – 74.
17. Owens DS, Parker PY, Benton D. Blood glucose and subjective energy following cognitive demand. Physiol Behav 1997;62 (3): 471 – 478.
18. Scholey AB, Harper S, Kennedy DO. Cognitive demand and blood glucose. Physiol Behav, 2001;73 (4), 585 – 592.
19. Leelayuwat N, Tunkumnerdthai O, Donsom M et al. An alternative exercise and its beneficial effects on glycaemic control and oxidative stress in subjects with type 2 diabetes. Diabetes Res Clin Pract, 2008;82, e5 – e8.
20. Ferrer-Garcia JC, Lopez PS, Pablos-Abella C et al. Benefits of a home-based physical exercise program in elderly subjects with type 2 diabetes mellitus. Endocrinol Nutr, 2011;58 (8), 387 – 394.
21. Labrunee M, Antoine D, Verges B et al. Effects of a home-based rehabilitation program in obese type 2 diabetics. Ann Rehabil Med, 2012;55 (6), 415 – 429.
22. Lin SP, Wang MJ. Strategic management of behavioural change in type 2 diabetic patients. Public Health,2012; 126 (1), 18 – 24.
23. Schutzer KA, Graves BS. Barriers and motivations to exercise in older adultes. Prev Med 2004;39(5), 1056-1061.
24. Ranawana V, Monro J.A, Mishra S, Henry K.J.C. Degree of particle size breakdown during mastication may be a possible cause of interindividual glycemic variability. Nutr Res 2010;30(4), 246-254.
25. Suzuki H, Fukushima M, Okamoto S et al. Effects of thorough mastication on postprandial plasma glucose concentrations in nonobese Japanese subjects. Metabolism 2005;54, 1593-1599.
26. Dekker MJ, Gusba JE, Robinson LE, Graham TE. Glucose homeostasis remains altered by acute caffeine ingestion following 2 weeks of daily caffeine consumption in previously non-caffeine-consuming males. Br J Nutr 2007;98 (3), 556 – 562.
27. O’Donovan G, Kearney EM, Nevill AM et al. The effects of 24 weeks of moderate- or high-intensity exercise on insulin resistance. Eur J Appl Physiol, 2005;95 (5-6), 522 – 528.
28. Koopman R, Manders RJF, Zorenc AHG et al. A single session of resistance exercise enhances insulin sensitivity for at least 24 h in healthy men. Eur J Appl Physiol, 2005;94 (1-2), 180 – 187.
