N. Luplertlop1, T. Kitisin2
1. Department of Microbiology and Immunology, Faculty of Tropical Medicine, Mahidol University, Bangkok, Thailand; 2. Department of Anatomy, Faculty of Science, Mahidol University, Bangkok, Thailand
Corresponding Author: Natthanej Luplertlop, Department of Microbiology and Immunology,
Faculty of Tropical Medicine, Mahidol University, Bangkok, Thailand. Tel: 66-894-807-337. E-mail: natthanej.lup@mahidol.ac.th
Abstract
Repetitive and long term exposure of solar UV irradiation can cause skin aging and induced wrinkling skin. These remarkable sequences are involving in accumulation of reactive oxygemspecies (ROS), induction of pro-inflammatory cytokines, and degradation of skin extreacellular matrix (ECM). Moreover, repetitive photo-aging by solar UV damaged together with chronological aging could cause significant changes in skin morphology. As the cosmetic market increasing, the high demands usage of synthetic anti-oxidative compounds consequently exhibited long term adverse effects to consumers. Thus, the attempt to find a natural plant extracts with anti-oxidative power can be used as a good candidate for anti-aging formulation. The present of this review mainly aimed to provide alternative plants information as a candidate to use their reported active ingredients, which exhibited possible potential on skin aging prevention. The review on these medicinal and edible plants with their constituents was described to highlight the potentials of the plants which may help to maximize the beneficial of anti-skin aging therapy.
Key words: Skin aging, photo-aging, wrinkling skin, photochemical compounds.
Introduction
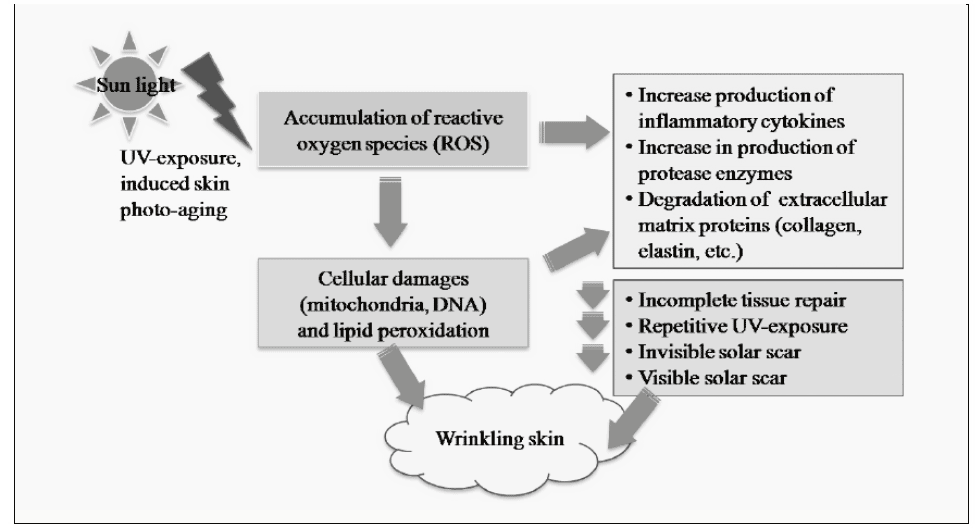
Aging refers to a remarkable complex event decreasing in health function and esthetic change of multiple organ systems that leads to increase mortality from stresses and diseases (1, 2). Moreover, the impact of aging on the function and appearance particular in the skin is recently growing interests. Among skin aging factors (both intrinsic and extrinsic), repetitive sunlight exposure has been considered to be the most possible factor that could cause skin aging (3). However, the precise physiological and biological mechanisms remain to be elucidated. Skin aging caused by UV exposure (photoaging) is superimposed on chronological skin aging. Photoaging and chronological skin aging showed various distinct entities in histology (4). Previous study reported that repetitive UV-exposure could cause skin aging and leaded to the accumulation of peroxyl free radicals, which caused from broken down of malondialdehyde (MDA) subsequently cross-links and polymerizes collagen. These events caused further decrased in skin elasticity water holding capacity in the skin, which a common and obvious symptom of photo-aged wrinkling skin (5). Thus, one of many pathways to prevent the aging effect on skin is due to scavenge the free radical formation in the skin by using anti-oxidative agents.
From the past, there are many cosmetic products that used chemical synthetic compounds that contained active ingredients including monoethanolamine, diethanolamine, sodium laureth sulfate, triethanolamine, etc, which may cause adverse effects such as skin allergy, contact dermaltitis, irritation, and photo-sensitive skin. Moreover, antioxidants such as vitamins A and E, squalene, co-enzyme Q10, ferulic acid, idebenone, pycnogenol and silymarin are being used in skin care products, which promoted skin repair from environmental damage to the skin (6). However, the cost of their pure compound is expensive. Because of this situation, the demands of skincare products that used natural plant extracts as anti-oxidative ingredients are growing interests. Searching a good candidate plant to find its active ingredient becomes challenge. Many medicinal plants and herbs, especially in the part of fruits, grains, and leaves are riches in anti-oxidant compounds that can scavenge free radicals, one of the reasons that can induce skin aging. In present, cosmetics produced from natural ingredients are now growing popular all over the world according to their significant positive effects on skin aging. This review article aimed to provide alternative plants information as a candidate to use their reported active ingredients in order to have some potential on skin aging prevention. The review on these plants and their constituents were described to highlight the potentials of the plants useful in skin aging from natural resources.
Plant extracts served as alternative anti- oxidative agents
Psidium guajava Linn
Psidium guajava Linn. (family Myrtaceae) has been that they are riches in flavonoids and phenolic compounds in leaves (16-18). The total phenolic content in the extract was deter- mined spectrophotometrically according to Folin–Ciocalteu’s phenol method and calculated as gallic acid equivalent (GAE). By using hot water extraction methods, dried leaves crude extracts exhibited remarkably high total phenolic content 575.3 ±
15.5 were obtained (19). Moreover, the antioxidant activity of lyophilized leaf extracts was determined using free radical DPPH (2,2-diphenyl-1-picrylhydryzyl) scavenging. It was showed that the leaves extracts of Psidium guajava Linn. exhibited similar trends of scavenging DPPH radical as ascorbic acid. Thus, it was concluded that the ability were from there phenolic contents such as protocatechuic acid, ferulic acid, quercetin and guavin B (7), quercetin, ascorbic acid, gallic acid and caffeic acid (20). Guava leaf extracts are a potential source of natural antioxidants (21).
Piper betel L.
Previous studies found active ingredients called allylpyrocatechol and chavibetol, which isolated from Piper betel L. leave extracts (22). However, allylpyrocatechol was found to be more potent. In addition, both active compounds exhibited protection of photosensitization-mediated lipid peroxidation of rat liver mitochondria (22). Moreover, Piper betel L. leave extracts showed a positive effect to prevent Cd induced oxidative hepatic dysfunction in rats. Prabu et al. concluded that the results may cause from allylpyrocatechol, which found in Piper betel L leaf (23). By using GC and GC-MS analysis. Prakash et al. revealed that there were 32 different components, which constitute 97% of the oil (8). In their investigation, eugenol (63.39%) and its ester derivative acetyleugenol (14.05%) were recorded as major components of oil. They also found that the anti-oxidative activity of the P. betle essential oil was found to be concentration dependent and very close to ascorbic acid, which implied that it save can an alternative source for anti-oxidative agent to synthetic anti-oxidants. These discoveries suggested that allylpyrocatechol and eugenol have an important role in protecting biological systems against cellular oxidative damage.
Schefflera leucantha R. Viguier
Schefflera leucantha R. Viguier, (Araliaceae), was found locally in Thailand and known as ‘Hanuman Prasankai’. Prevoius reported indicatied that Schefflera leucantha R. Viguier crude extracts exhibited saponins (24). This phytochemical active compound showed an anti-oxidative activity similar to ascorbic acid on DPPH radical scavenging assay (9). Moreover, an aqueous Schefflera leucantha R. Viguier leave extracts were described as hypoglycemic activity in rats and anti-fungal activity (9, 25). From these study, it was concluded that the leaves of S. leucantha had benefit for extracting its crude oil to develop as an anti-oxidants.
Andrographis paniculata Nees
Andrographis paniculata Nees (A. paniculata) is commonly known as Creat or King of bitters due to its bitter taste, and it was often called as Fah Tah Lai in Thailand. From the past, crude aqueous extract of A. paniculata has long been used to treat diarrhoea with overall effectiveness of 91.3% (26). Rafat et al. reported that the crude extracts of A. paniculata exhibited anti- oxidant activity due to it contained phenolic compounds (27). Furthermore, in 2011, Wasman et al. concluded that the ethanol extract exhibited more phenolic content and presented stronger activity than aqueous extract (28). Moreover, it was found that the high phenolic contents were identified as andrographolide (AND) and echiodinin (ECH), which exhibited postitive scavenging result on DPPH assay when compared with ascorbic acid (10). Moreover, from Arifullah et al. study, the andrographolide (AND) and echiodinin (ECH) purified from A. paniculata crude extracts by using thin layer chromatography methods showed anti-bacterial activity in both gram postitive and gram negative bateria including avirulent Mycobacterium smegmatis (10).
However, they also found that the anti-oxidative activity from purified echiodinin (ECH) showed higher activity in as compared to purified andrographolide (AND). Thus, the higher anti-oxidant activity in purified echiodinin (ECH) may cause from their number and position of free hydroxyl group attached to phenol rings that can readily donates the phenolic hydrogens or electron to the acceptor molecules (29).
Garcinia mangostana Linn
Garcinia mangostana Linn or commonly known as mangosteen is one of edible fruit in Thailand. The fruit of mangosteen is dark purple or reddish peel, with internal white, soft and juicy edible pulp. The pulp contained a slightly acid, sweet flavor, and a pleasant aroma (30). Interestingly, Xanthones or xanthen-9H-ones are secondary metabolites that found most in Garcinia mangostana Linn. An α-mangostin, as known as active compound was firstly xanthone isolated in Garcinia mangostana Linn. pericarp (31) and later were ♌-mangostin (32) and γ -mangostin (33). Moreover, xanthones derivative compounds also found in many parts of the G. mangostana such as fruit, bark, and leaves (34). In the study of anti-oxidant activity of extracts and xanthones isolated from Garcinia mangostana Linn by using DPPH assay revealed that ♋-mangostin exhibited an ability to scarvenage DPPH readical (11). Moreover, ♋-mangostin was found to reduce human low density lipoproteins (LDL) oxidation induced by copper or peroxyl radical (35), prevented the decrease of the ♋-tocopherol consumption induced by LDL oxidation (36), induced apoptosis in human leukemia cell lines (37), and exhibited anti-tumoral activity against DLD-1 cells (38). These findings indicated that usage of the anti-oxidant properties of extracts from Garcinia mangostana Linn.
Oryza sativa L.
Rice (Oryza sativa L.) is one of the most important edible crops in Thailand. From the past, many studies have reported several active ingredients from rice, e.g. gamma-oryzanol (γ-oryzanol) (39), tocopherols, and tocotrienols (40). In addition, rice bran is a potential source of various discovered active compounds (41). Gamma-oryzanol is a complex mixture of ferurate esterfied with sterols or triterpene alcohols (42) and was purposed to be the major anti-oxidant compound found in rice bran (43). Moreover, crude rice oil extracted from rice grain had been used to study anti-oxidative property by using DPPH assay and HPLD analysis. Butsat and Siriamornpun showed that Oryza sativa L. Thai rice called Khao Dawk Mali 105 exhibited anti-oxidative property and showed high contents of γ-oryzanol and tocopherols found in barn (44). They also found three major phenolic acids, which were ferulic acid (found most evident in the bran), vanillic, and p-coumaric acids (found mostly in the husk). Another types of rice is coloured rice, particularly black rice (Oryza sativa L. indica), which contained natural anthocyanin compounds, such as cyanidin 3-glucoside and peonidin 3-glucoside, which exhibited anti-oxidative activities (45). Together with γ-oryzanol, the black rice crude extracts showed a reduction of reactive oxygen species (ROS) such as lipid peroxide and superoxide anion radicals and reduced cholesterol content in hypercholesterolemic rats (46, 47).
Recently, our original studies have been conducted to study the anti-oxidative activity in crude rice extracts as an attempt to find an alternative way to utilize cadmium- contaminated rice without introducing health hazard risks. From the past, it was discovered in Thailand that there is a cadmium contamination occurred in rice and soil in Mae Sot District, Tak Province (48). However, biotic and abiotic stress, such as cadmium stress, induced plant to product secondary metabolite products as defense mechanisms against the stress (49). Our studies showed that crude rice oil extracted from cadmium- contaminated rice exhibited anti-oxidative property as determined by using DPPH assay (15). We also found that the main active compound found in crude cadmium- contaminated rice oil was a γ-oryzanol as determined by using RP-HPLC. It was found that the γ-oryzanol content in crude rice oil extracted from cadmium-contaminated rice was higher than control crude rice oil. Thus, it was implied that higher anti-oxidative compound found in crude cadmium-contaminated rice oil may cause from the defense mechanisms of secondary metabolites production in rice in different levels both biotic and abiotic stress. Moreover, the crude rice oil extracted from cadmium- contaminated rice contained very low level of cadmium concentration, which considered safe for consumption (15). We also determined the anti-oxidative activity of crude rice oil in an in vitro primary human skin fibroblast cell model. The results showed that crude rice oil extracted from cadmium-contaminated rice reduced oxidative damage on cellular mitochondrial activity, exhibited down-regulation of SIRT1 mRNA, and up- regulation of MMP-2 in PHF cells in vitro, which may promote tissue remodeling (14). Our studies showed significant information of crude rice oil extracted from cadmium-contaminated rice, which exhibited the potential of anti-oxidative activity and can be utilized without compromising human health with hazard risks.
Conclusion
The information of this review provided a significant scientific validity on usage of medicinal and edible plant extracts as an anti-oxidiative agent, which should be investigated further based on different models. Moreover, from previous studies, it was amazingly that these reviewed plants also showed another effects such as anti- inflammatory, anti-fungal, anti-cancer effects, etc. However, there are lots of experiments need to be studied in depth. It is still very clear that all these reviewed plants are goods promising candidates for serving as therapeutic substances or cosmeceutical potential in the future. Nevertheless, further studies need to be done in order to investigate the effects of these plants extracts in animal models in vivo and/or in human.
References
-
Rabe JH, Mamelak AJ, McElgunn PJ, Morison WL, Sauder DN. Photoaging: mechanisms and repair. J Am Acad Dermatol 2006;55:1-19
-
Haigis MC, Sinclair DA. Mammalian sirtuins: biological insights and disease relevance. Annu Rev Pathol 2010;5:253-295
-
itti L isher ( ) -light-induced signal cascades and skin aging. Ageing Res Rev 1:705-720
-
Lavker RM. Cutaneous aging: chronologic versus photoaging. In: Gilchrest B (ed) Photodamage, Vol. 1. Blackwell, Cambridge,1995; pp 123-135
-
Kim YH, Kim KS, Han CS et al. Inhibitory effects of natural plants of Jeju Island on elastase and MMP-1 expression. J Cosmet Sci 2007;58:19-33
-
Pinnell SR. Cutaneous photodamage, oxidative stress, and topical antioxidant protection. J Am Acad Dermatol 2003;48:1–19
-
Thaipong K, Boonprakob U, Cisneros-Zevallos L, Byrne DH. Hydrophilic and lipophilic antioxidant activities of guava fruits. Southeast Asian J Trop Med Public Health 2005;36:254–257
-
Prakash B, Shukla R, Singh P, Kumar A, Mishra PK, Dubey NK. Efficacy of chemically characterized Piper betle L. essential oil against fungal and aflatoxin contamination of some edible commodities and its antioxidant activity. Int J Food Microbiol 2010;15:114–119
-
Potduang B, Chongsiriroeg C, Benmart Y, Giwanon R, Supatanakul W, Tanpanich S. Biological Activities of Schefflera Leucantha. Afr J Tradit Complement Altern Med 2007;4:157–164
-
Arifullah M, Namsa ND, Mandal M, Chiruvella KK, Vikrama P, Gopa GR. Evaluation of anti-bacterial and anti-oxidant potential of andrographolide and echiodinin isolated from callus culture of Andrographis paniculata Nees. Asian Pac J Trop Biomed 2013;3:604-610
-
Yoshikawa M, Harada E, Miki A et al. Antioxidant constituents from the fruit hulls of mangosteen (Garcinia mangostana L.) originating in Vietnam. Yakugaku Zasshi 1994;114:129–133
-
Ho CK, Huang YL, Chen CC. Garcinone E, a xanthone derivative, has potent cytotoxic effect against the hepatocellular carcinoma cell lines. Planta Med 2002;68:975–979
-
Chen LG, Yang LL, Wang CC. Anti-inflammatory activity of mangostins from Garcinia mangostana. Food Chem Toxicol 2008;46:688–693
-
Kitisin T, Kosiyachinda P, Luplertlop N (2013) Potential anti-oxidative activity of crude rice oil extracted from cadmium-contaminated rice as determined using an in vitro primary human fibroblast cell model. J Agri Sci 2013;5:104-121
-
Kitisin T, Visoottiviseth P, Drechsel H, Kosiyachinda P, Luplertlop N (2013) Anti-oxidative property of crude rice oil extracted from cadmium- contaminated rice. J Environ Sci Health, 2013;Part B 48:823–829
-
Li J, Chen F, Luo J (1999) GC–MS analysis of essential oil from the leaves of Psidium guajava. Zhong Yao Cai 1999;22:78–80
-
Vargas AD, Soto HM, Gonzalez HVA, Engleman EM, Martinez GA (2006) Kinetics of accumulation and distribution of flavonoids in guava (Psiduim guajava). Agrociencia 2006;40:109–115
-
Gutiérrez RM, Mitchell S, Solis RV. Psidium guajava: a review of its traditional uses, phytochemistry and pharmacology. J Ethnopharmacol 2008;117:1-27
-
Qian H, Nihorimbere V. Antioxidant power of phytochemicals from Psidium guajava leaf. J Zhejiang Univ Sci 2004;5:676–683
-
Jiménez-Escrig A, Rincón M, Pulido R, Saura-Calixto F. Guava fruit (Psidium guajava L.) as a new source of antioxidant dietary fiber. J Agric Food Chem 2001;49:5489–5493
-
Soman S, Rauf AA, Indira M, Rajamanickam C. Antioxidant and antiglycative potential of ethyl acetate fraction of Psidium guajava leaf extract in streptozotocin-induced diabetic rats. Plant Foods Hum Nutr 2010;65:386-391
-
Mula S, Banerjee D, Patro BS et al. Inhibitory property of the Piper betel phenolics against photosensitization-induced biological damages. Bioorg Med Chem 2008;16:2932–2938
-
Prabu SM, Muthumani M, Shagirtha K. Protective effect of Piper betle leaf extract against cadmium-induced oxidative stress and hepatic dysfunction in rats. Saudi J Biol Sci 2012;19:229–239
-
Pancharoen O, Tuntiwachwuttikul P, Taylor WC, Picker K. Triterpenoid glycosides from Schefflera lucantha. Phytochemistry 1994;35:987-992
-
Satayavivad J, Bunyapraphatsara N, Thiantanawat A, Kositchaiwat U. Hypoglycemic activity of the aqueous extract of Schefflera leucantha Viguier in rats. Thai J Phytopharmacy 1996;3:1-5
-
Chopra RN, Nayar SL, Chopra IC. Glossary of Indian medicinal plants. Council of Scientific and Industrial Research, New Delhi, 1980.
-
Rafat A, Koshy P, Sekaran M. Antioxidant potential and content of phenolic compounds in ethanolic extracts of selected parts of Andrographis paniculata. J Med Plants Res 2010;4:197-202
-
Wasman SQ, Mahmood AA, Chua LS, Alshawsh MA, Hamdan S. Antioxidant and gastroprotective activities of Andrographis paniculata (Hempedu Bumi) in Sprague Dawley rats. Indian J Exp Biol 2011;49:767-772
-
Arteaga JF, Ruiz-Montoya M, Palma A, Alonso-Garrido G, Pintado S, Rodríguez-Mellado JM. Comparison of the simple cyclic voltammetry (CV) and DPPH assays for the determination of antioxidant capacity of active principles. Molecules 2012;17:5126-5138
-
Jung HA, Su BN, Keller WJ, Mehta RG, Kinghorn AD. Antioxidant Xanthones from pericarp of Garcinia mangostana (Mangosteen). J Agric Food Chem 2006;54:2077–2082
-
Schmid W. Ueber das mangostin. Liebigs Ann Chem 1855;93:83–89
-
Dragendorff O. er das Har von arcinia angostana L. Lie igs nn 1930;482:280–301
-
Jefferson AQA, Scheimann F, Sim KY. Isolation of c-mangostin from Garcinia Mangostana and preparation of the natural mangostins by selective demethylation. Aust J Chem 1970;23:2539–2543
-
Pedraza-Chaverri J, Cárdenas-Rodríguez N, Orozco-Ibarra M, Pérez-Rojas JM; Medicinal properties of mangosteen (Garcinia mangostana). Food Chem Toxicol 2008;46:3227–3239
-
Williams P, Ongsakul M, Proudfoot J, Croft K, Beilin L. Mangostin inhibits the oxidative modification of human low density lipoprotein. Free Radic Res 1995;23:175–184
-
Mahabusarakam W, Proudfoot J, Taylor W, Croft K (2000) Inhibition of lipoprotein oxidation by prenylated xanthones derived from mangostin. Free Radic Res 200;33:643–659
-
Matsumoto K, Akao Y, Ohguchi K et al. Xanthones induce cell-cycle arrest and apoptosis in human colon cancer DLD-1 cells. Bioorg Med Chem 2005;13:6064–6069
-
Nakagawa Y, Iinuma M, Naoe T, Nozawa Y, Akao Y. Characterized mechanism of -mangostin-induced cell death: Caspase-independent apoptosis with release of endonuclease-G from mitochondria and increased miRNA-143 expression in human colorectal cancer DLD-1 cells. Bioorg Med Chem 2007;15:5620–5628
-
Xu Z, Godber JS. Purification and identification of components of γ-oryzanol in rice bran oil. J Agric Food Chem 1999;47:2724-2728
-
Tai-sun S, Godber JS. Isolation of four tocopherols and four tocotrienols from variety of natural sources by semi-preparative high-performance liquid chromatography. J Chromatogr A 1994;678:49-58
-
Nicolosi RJ, Rogers EJ, Ausman LM, Orthoefer FT. Rice bran oil and its health benefits. In: Marshall WE, Wadsworth JI (eds) Rice science and technology. Marcel Dekker, New York, 1994;pp 350–421
-
Rogers EJ, Rice SM, Nicolosi RJ, Carpenter DR, McClelland CA, Romanczyk LJ Jr. Identification and quantitation of γ–oryzanol components and simultaneous assessment of tocopherols in rice bran oil. J Am Oil Chem Soc 1993;70:301-307
-
Xu Z, Godber JS. ntioxidant activities of major components of γ-oryzanol from rice bran using a linoleic acid model. J Am Oil Chem Soc 2001;78:645- 649
-
Butsat S, Siriamornpun S. Antioxidant capacities and phenolic compounds of the husk, bran and endosperm of Thai rice. Food Chem 2010;119:606–613
-
Hu C, Zawistowski J, Ling W, Kitts DD. Black rice (Oryza sativa L. indica) pigmented fraction suppresses both reaction oxygen species and nitric oxide in chemical and biological model systems. J Agric Food Chem 2003;51:5271–5277
-
Ichikawa H, Ichiyanagi T, Xu B, Yoshii Y, Nakajima M, Konishi T. Antioxidant activity of anthocyanin extract from purple black rice. J Med Food 2001;4:211–218
-
Nam YJ, Nam SH, Kang MY. Cholesterol-lowering efficacy of unrefined bran oil from the pigmented black rice (Oryza sativa L cv. Suwon 415) in hypercholesterolemic rats. Food Sci Biotechnol 2008;17:457–463
-
Simmons RW, Pongsakul P, Saiyasitpanich D, Klinphoklap S. Elevated levels of cadmium and zinc in paddy soils and elevated levels of cadmium in rice grain downstream of a zinc mineralized area in Thailand: implications for public health. Environ Geochem Health 2005;27:501–511
-
Namdeo AG. Plant cell elicitation for production of secondary metabolites: a review. Phcog Rev 2007;1:69-79